Dr. Shanti Swarup, Adjunct Professor, Coatings and Polymeric Materials, North Dakota State University; PPG Collegium Member02.16.22
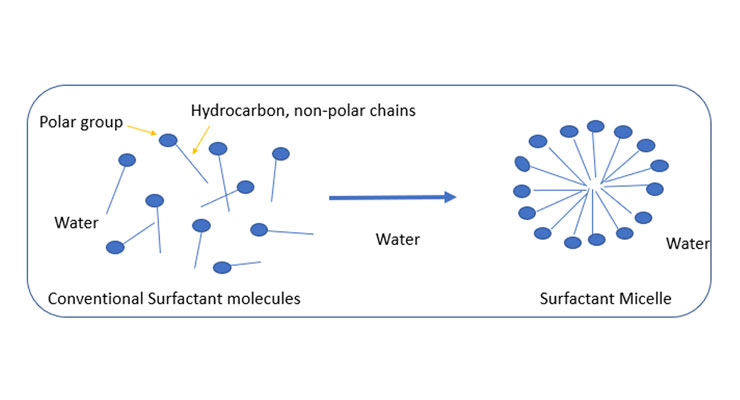
Conventional surfactants are molecules that include both polar and non-polar groups in the same molecules that are able to change the properties of the liquid in which they are solubilized. They are low molecular weight (<500 g mol−1) molecules and start to form aggregates in water known as micelles.
The surfactant concentration at which these micelles are formed is known as critical micellar concentration (CMC). The non-polar hydrophobic groups are oriented inwards in the micelles forming a hydrophobic environment. The polar groups reside in contact with water. Depending on the nature of the polar groups, they can be classified as anionic, cationic, non-ionic or zwitterionic1.
One of the important properties of surfactants is that they solubilize non-polar molecules in these micelles, which serves the basis for synthesizing waterborne polymers known as latexes. The amount of surfactant used should be more than their CMC. These latexes are widely used in many industrial applications such as biotechnology, coatings, medical applications, personal care, water treatment and enhanced oil recovery. However, surfactants, due to their low molecular weight and hydrophilic groups, absorb moisture in the coating films, causing defects such as corrosion, blistering, adhesion failure, and acid etching.
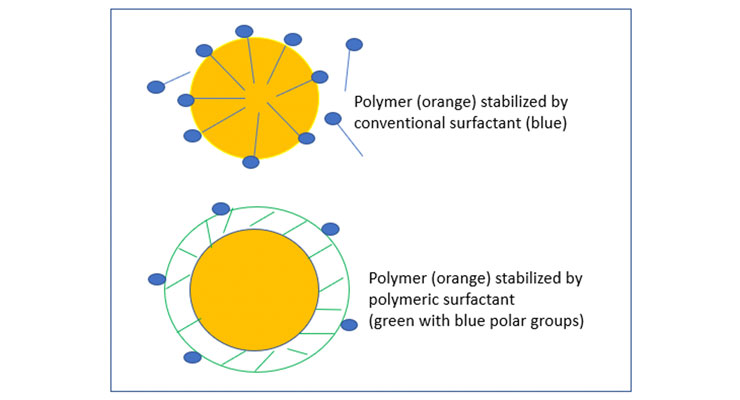
Unlike conventional surfactants, polymeric surfactants are of much higher molecular weight, thousands to tens of thousands, and their polar groups are distributed over their backbone, or at the both side of non-polar chain. The polymeric surfactants of high hydrophobicity, high molecular weight, and low polarity can overcome defects associated with conventional surfactants. They can provide additional advantages such as building rheology during application and encapsulating materials.
Here I describe couple of examples of polymeric surfactants.
1. Polymeric surfactants based on urethane backbone: The production of polyurethane surfactants include the reaction between isocyanate groups and hydroxyl groups leading to urethane groups.
a. Anionic Polyurethane Surfactants: Anionic polyurethane surfactant (APUS) can be prepared by reacting di- or tri- isocyanate, polyol, and acid containing diol such as di-methalol propionic or butanoic acid. Stoichiometry should be chosen such that hydroxyl groups are in excess than isocyanate. The acid value based on the total polymer solid should be in the range of 20-40. These materials can be prepared in an inert solvent such as methyl ethyl ketone (MEK) at around 80°C using tin or tertiary amine as a catalyst.
The material can then be inverted into deionized water using base such as dimethyl ethanol or triethyl amine as a neutralizing base for the acid groups. The degree of neutralization can be adjusted to control the solid-viscosity of the finished product. Solvent can be distilled off to produce solvent free product.
Zheng and Lu2 prepared anionic polyurethane surfactant (APUS) by reacting isophorone diisocyanate, polyethylene glycol 400, 2,2-bis(hydroxymethyl)propionic acid and octanol at 80°C using dibutyl tin dilaurate as a catalyst. The material they prepared exhibited the following properties: Mn (number average molecular weight) = 3232 g mol−1; CMC =
2.04 mmol L−1; surface tension at CMC = 35.28 mN m−1. Several latexes were prepared using 2% of this APUS with varying amounts of Methyl methacrylate (MMA), butyl acrylate (BA) monomers and potassium persulfate (KPS) as free radical forming initiator. The latexes made from APUS were compared to the latexes made with conventional low molecular weight surfactant, sodium dodecyl sulfate (SDS). It was found that APUS based latexes were much superior for emulsion stability, water resistance and mechanical properties (tensile strength and elongation). Tailor-made APUS can be made using other polyols and isocyanates to achieve desired properties which cannot be achieved with conventional surfactants.
b. Non-ionic Polyurethane Surfactants: Non-ionic polyurethane surfactants (NPUS) can be made by reacting isocyanates with polyols or hydroxyl amines, or both similar to the process described above. Negm et. al3 prepared several NPUS by reacting polyethylene glycol of different molecular weights with toluene diisocyanate. The average molecular weights of these polyurethanes were controlled in the range 3900-100,000 g/mol. The CMC values of these materials were comparable to conventional non-ionic low molecular weight commercially available surfactants such Tween-20, BRIJ 35, and Triton X-10. The particle size ranged 30 nm-140nm, longer the ethylene glycol chain, shorter the particle size. The latexes made from these surfactants are freeze-thaw stable and can provide excellent flexibility in the coating applications.
2. Polymeric surfactants based on the acrylic backbone: There are two broad groups of these surfactants. One group contains random copolymers of acrylic acid or methacrylic acid with hydrophobic monomers such as styrene, butyl acrylate, or octyl acrylate prepared in organic solvents by free radical polymerization followed by neutralization with base and then inversion in water. The solvent can be removed by distillation. Latexes with good colloidal stability can be prepared using these polymeric surfactants4.
The other group is styrene-maleic anhydride (SMA) copolymers followed by ring opening of anhydride with alcohol. SMA surfactants obtained by ring opening with long fatty alcohols, C10 or more were successfully prepared and used to make latexes5. High solid latexes (solid 40%) with good stability were made when SMA copolymers were reacted with dodecanol compared to SMA reacted with butanol. Incorporation of long alkyl chain not only decreases the acid number but also increases hydrophobic portion, providing better hydrophilic-lipophilic balance, offering the latexes both steric as well as charge stabilization.
These surfactants can be used in higher amount (10-20%) to copolymerize hydrophobic monomers such as butyl acrylate, styrene, isobornyl acrylate and can optionally have functional monomers such as hydroxy ethyl methacrylate for crosslinking in the coatings. Latexes made from these surfactants have core: shell type morphology. The chains in the core of these latexes can be crosslinked using di or triacrylate such as hexanediol diacrylate or triallyl cyanurate. Crosslinked latexes are useful in minimizing or preventing interlayer mixing.
The polymeric surfactants due to their much higher molecular weight, long flexible polymeric chain higher particle size can offer several advantages over conventional surfactants. They can be easily made in the laboratory based on the desired chemistries and properties needed for applications.
References:
1S. Swarup; C. K. Schoff; A Survey of Surfactants in Coatings Technology; Progress in Organic Coatings; vol. 23. p. 1-22 (1993)
2Jie Zheng; Man-Geng Lu; A Polyacrylate Prepared using Polyurethane Surfactants, Chemical Papers, vol. 69, p. 709–715 (2015)
3Nabel A. Negm; Maher A. El Hashash; Mona A. Youssif; Eid A. Ismail; Zizi I. Abdeen; Nasser R. Abdel Rahman; Novel Nonionic Polyurethane Surfactants and Ag Nanohybrids: Influence of Nonionic Polymeric Chains; J Surfact Deterg vol. 20, 173–182 (2017)
4Muli Wang; Zhijun Ma; Dong Zhu; Dongyang Zhang; Wu Yin; Core-shell latex synthesized by emulsion polymerization using an alkali-soluble resin as sole surfactant; Journal of Applied Polymer Sci., vol 128, p. 4224-4223 (2013)
5Zhang, Huaihao; Bai, Yongqing; Zhao, Jing; Shi, Qiaofang; Zang, Yang; Wu, Jun; Designing, Synthesizing, and Analyzing a Comb-like Polymeric Surfactant, Poly (acrylic acid-co-octadecyl acrylate); Journal of chemical education, 2021-06-08, Vol. 98, p. 2074-2082 (2021)
Dr. Swarup is an Adjunct Professor in the department of Polymeric Materials and Coatings, North Dakota State University. His research interests are in polymers, surfactants and additives synthesis and their coating applications. He received his Ph.D. from Aligarh Muslim University, India and Postdoctoral Fellowships from Lund, and Uppsala University (Sweden), and Rutgers University. He joined PPG Industries in 1988 and retired as Associate Fellow in 2020. During his stay as a polymer scientist at PPG, he and his team developed and scaled-up 220 unique polymers. Fifty-five of these polymers are used in commercial coating products. He is named inventor on >700 global patents and publications including 97 granted US patents and over 25 pending review in the U.S. patent office.
The surfactant concentration at which these micelles are formed is known as critical micellar concentration (CMC). The non-polar hydrophobic groups are oriented inwards in the micelles forming a hydrophobic environment. The polar groups reside in contact with water. Depending on the nature of the polar groups, they can be classified as anionic, cationic, non-ionic or zwitterionic1.
One of the important properties of surfactants is that they solubilize non-polar molecules in these micelles, which serves the basis for synthesizing waterborne polymers known as latexes. The amount of surfactant used should be more than their CMC. These latexes are widely used in many industrial applications such as biotechnology, coatings, medical applications, personal care, water treatment and enhanced oil recovery. However, surfactants, due to their low molecular weight and hydrophilic groups, absorb moisture in the coating films, causing defects such as corrosion, blistering, adhesion failure, and acid etching.
Unlike conventional surfactants, polymeric surfactants are of much higher molecular weight, thousands to tens of thousands, and their polar groups are distributed over their backbone, or at the both side of non-polar chain. The polymeric surfactants of high hydrophobicity, high molecular weight, and low polarity can overcome defects associated with conventional surfactants. They can provide additional advantages such as building rheology during application and encapsulating materials.
Here I describe couple of examples of polymeric surfactants.
1. Polymeric surfactants based on urethane backbone: The production of polyurethane surfactants include the reaction between isocyanate groups and hydroxyl groups leading to urethane groups.
a. Anionic Polyurethane Surfactants: Anionic polyurethane surfactant (APUS) can be prepared by reacting di- or tri- isocyanate, polyol, and acid containing diol such as di-methalol propionic or butanoic acid. Stoichiometry should be chosen such that hydroxyl groups are in excess than isocyanate. The acid value based on the total polymer solid should be in the range of 20-40. These materials can be prepared in an inert solvent such as methyl ethyl ketone (MEK) at around 80°C using tin or tertiary amine as a catalyst.
The material can then be inverted into deionized water using base such as dimethyl ethanol or triethyl amine as a neutralizing base for the acid groups. The degree of neutralization can be adjusted to control the solid-viscosity of the finished product. Solvent can be distilled off to produce solvent free product.
Zheng and Lu2 prepared anionic polyurethane surfactant (APUS) by reacting isophorone diisocyanate, polyethylene glycol 400, 2,2-bis(hydroxymethyl)propionic acid and octanol at 80°C using dibutyl tin dilaurate as a catalyst. The material they prepared exhibited the following properties: Mn (number average molecular weight) = 3232 g mol−1; CMC =
2.04 mmol L−1; surface tension at CMC = 35.28 mN m−1. Several latexes were prepared using 2% of this APUS with varying amounts of Methyl methacrylate (MMA), butyl acrylate (BA) monomers and potassium persulfate (KPS) as free radical forming initiator. The latexes made from APUS were compared to the latexes made with conventional low molecular weight surfactant, sodium dodecyl sulfate (SDS). It was found that APUS based latexes were much superior for emulsion stability, water resistance and mechanical properties (tensile strength and elongation). Tailor-made APUS can be made using other polyols and isocyanates to achieve desired properties which cannot be achieved with conventional surfactants.
b. Non-ionic Polyurethane Surfactants: Non-ionic polyurethane surfactants (NPUS) can be made by reacting isocyanates with polyols or hydroxyl amines, or both similar to the process described above. Negm et. al3 prepared several NPUS by reacting polyethylene glycol of different molecular weights with toluene diisocyanate. The average molecular weights of these polyurethanes were controlled in the range 3900-100,000 g/mol. The CMC values of these materials were comparable to conventional non-ionic low molecular weight commercially available surfactants such Tween-20, BRIJ 35, and Triton X-10. The particle size ranged 30 nm-140nm, longer the ethylene glycol chain, shorter the particle size. The latexes made from these surfactants are freeze-thaw stable and can provide excellent flexibility in the coating applications.
2. Polymeric surfactants based on the acrylic backbone: There are two broad groups of these surfactants. One group contains random copolymers of acrylic acid or methacrylic acid with hydrophobic monomers such as styrene, butyl acrylate, or octyl acrylate prepared in organic solvents by free radical polymerization followed by neutralization with base and then inversion in water. The solvent can be removed by distillation. Latexes with good colloidal stability can be prepared using these polymeric surfactants4.
The other group is styrene-maleic anhydride (SMA) copolymers followed by ring opening of anhydride with alcohol. SMA surfactants obtained by ring opening with long fatty alcohols, C10 or more were successfully prepared and used to make latexes5. High solid latexes (solid 40%) with good stability were made when SMA copolymers were reacted with dodecanol compared to SMA reacted with butanol. Incorporation of long alkyl chain not only decreases the acid number but also increases hydrophobic portion, providing better hydrophilic-lipophilic balance, offering the latexes both steric as well as charge stabilization.
These surfactants can be used in higher amount (10-20%) to copolymerize hydrophobic monomers such as butyl acrylate, styrene, isobornyl acrylate and can optionally have functional monomers such as hydroxy ethyl methacrylate for crosslinking in the coatings. Latexes made from these surfactants have core: shell type morphology. The chains in the core of these latexes can be crosslinked using di or triacrylate such as hexanediol diacrylate or triallyl cyanurate. Crosslinked latexes are useful in minimizing or preventing interlayer mixing.
The polymeric surfactants due to their much higher molecular weight, long flexible polymeric chain higher particle size can offer several advantages over conventional surfactants. They can be easily made in the laboratory based on the desired chemistries and properties needed for applications.
References:
1S. Swarup; C. K. Schoff; A Survey of Surfactants in Coatings Technology; Progress in Organic Coatings; vol. 23. p. 1-22 (1993)
2Jie Zheng; Man-Geng Lu; A Polyacrylate Prepared using Polyurethane Surfactants, Chemical Papers, vol. 69, p. 709–715 (2015)
3Nabel A. Negm; Maher A. El Hashash; Mona A. Youssif; Eid A. Ismail; Zizi I. Abdeen; Nasser R. Abdel Rahman; Novel Nonionic Polyurethane Surfactants and Ag Nanohybrids: Influence of Nonionic Polymeric Chains; J Surfact Deterg vol. 20, 173–182 (2017)
4Muli Wang; Zhijun Ma; Dong Zhu; Dongyang Zhang; Wu Yin; Core-shell latex synthesized by emulsion polymerization using an alkali-soluble resin as sole surfactant; Journal of Applied Polymer Sci., vol 128, p. 4224-4223 (2013)
5Zhang, Huaihao; Bai, Yongqing; Zhao, Jing; Shi, Qiaofang; Zang, Yang; Wu, Jun; Designing, Synthesizing, and Analyzing a Comb-like Polymeric Surfactant, Poly (acrylic acid-co-octadecyl acrylate); Journal of chemical education, 2021-06-08, Vol. 98, p. 2074-2082 (2021)
Dr. Swarup is an Adjunct Professor in the department of Polymeric Materials and Coatings, North Dakota State University. His research interests are in polymers, surfactants and additives synthesis and their coating applications. He received his Ph.D. from Aligarh Muslim University, India and Postdoctoral Fellowships from Lund, and Uppsala University (Sweden), and Rutgers University. He joined PPG Industries in 1988 and retired as Associate Fellow in 2020. During his stay as a polymer scientist at PPG, he and his team developed and scaled-up 220 unique polymers. Fifty-five of these polymers are used in commercial coating products. He is named inventor on >700 global patents and publications including 97 granted US patents and over 25 pending review in the U.S. patent office.