Shiying Zheng, Shafiq Fazel, and Robert Rasing, Evonik Corporation and Evonik Resource Efficiency GmbH08.02.21
Abstract
Two-component epoxy technology provides excellent chemical resistance, mechanical strength and adhesion to various substrates. It has been widely used in many different applications, especially in concrete coatings. Traditionally, many epoxy systems require the use of plasticizer to ensure full chemical conversion of the polymer matrix. One of the challenges from epoxy floor coating application is the potential emission of plasticizer to the building inhabitants and environment throughout the service life of a floor. Legislation and end-user demand have driven new product development to focus on mitigating potential emissions from the plasticizer, as well as maintaining high performance.
Epoxy concrete coatings are essential to improve aesthetics, durability and provide protection to concrete substrates. Another challenge for concrete floor coatings is the moisture vapor transmission through the concrete floor. Moisture can cause deterioration of flooring adhesives and delamination of coatings by osmotic action, thus reducing or eliminating the benefits of epoxy coatings on concrete. It is one of the frequent causes for floor failure. The cost to replace a failed floor can be significant. Recent innovative technology developments have offered a solution to remediate the issue of moisture vapor transmission through the concrete floor.
This paper will present epoxy systems based on innovative curing agent technology that contains very low to no plasticizer and has excellent moisture vapor barrier property as thin coating. Fundamental study highlights the full property development of these systems. The paper will discuss the key performance attributes using these new curing agent systems. A combination of high barrier property to moisture vapor, low to no plasticizer, tailorable working time, and fast property development offers coating formulators the choices to design new floor systems incorporating thin epoxy coatings as effective moisture vapor barrier to meet specific requirements.
Introduction
Concrete is one of the most used construction materials due to its strength, durability, resilience, safety, and low cost. In flooring applications, demand for concrete for both interior and exterior use in residential and commercial settings is growing significantly thanks to the strong construction market around the globe. This growth includes new housing jobs as well as refurbishing and remodeling work. Concrete coatings have multiple functions where they can serve as an impermeable barrier, protect concrete from wear, deterioration, and contamination, enhance physical performance, provide chemical resistance, and improve aesthetics. An impermeable barrier is essential for concrete floorings. Since concrete is a permeable material, it allows not only chemicals from above to permeate and attack the concrete, but also the moisture originating from below the concrete or trapped in the concrete itself, to migrate as a vapor phase to concrete surface. Cured concrete traps varied amount of moisture, about 1%-2% in ambient dry concrete, and 4% to 5% in wet concrete.1
Moisture transfer phenomenon accounts for many flooring failures resulting in significant financial losses annually. Excessive moisture leads to damaged floor coverings, safety hazards due to slips and falls, and microbial growth leading to reduced indoor air quality. A moisture vapor barrier coating is proven to be an effective solution to address flooring failures by minimizing moisture vapor transmission. Applicators can apply a moisture vapor barrier coating for both existing and new construction jobs before applying the resilient floor covering, such as tile, wood, carpet, laminate, or installing a seamless flooring system of multiple coating layers for example broadcast floors, or Terrazzo.
Well-designed epoxy floor coatings are often used as moisture vapor barrier due to their excellent barrier property. Many epoxy systems require the use of reactive epoxy diluent, or plasticizer such as benzyl alcohol to ensure full chemical conversion of the polymer matrix by reducing the concentration of reactive groups and increasing mobility of the reactive species.2 These approaches enable a sufficient degree of cure well beyond the gel point to provide high performance of the flooring product.3 One of the challenges from epoxy floor coatings application is the potential emission of plasticizer to the building inhabitants and environment throughout the service life of a floor. One area of the new product development for epoxy floor coatings is to minimize the potential emissions from the plasticizer.
The primary objective of this paper is to present the development of new curing agent technology that contains very low to no plasticizer content and has excellent moisture vapor barrier properties as thin coating. The paper will include the comparison study of the new technology versus the conventional plasticizer containing products and the key performance benefits using the new curing agents. The new curing agent systems display a high degree of cure at ambient and low temperature, excellent moisture vapor barrier properties, and improved resistance to mechanical stress and temperature exposures.
Experimental
The new amine curing agents used in this study as moisture vapour barrier are represented as MVB1, MVB2, and MVB3, and the benchmarking conventional curing agents as CA1 and CA2.4 All coatings or castings were prepared with standard bisphenol A liquid epoxy resin or bisphenol A/F epoxy resin diluted with a glycidyl ether of C12-14 alcohol at 1:1 stoichiometry unless otherwise specified.5
The viscosity profiles were obtained on a Brookfield viscometer at 25oC using about 12 grams of mixed material. Coatings for thin film set time and Persoz hardness were deposited on glass substrates at 150 µm wet film thickness (WFT). The thin film set time (TFST) was determined using a Beck-Koller recorder, in accordance with ASTM D5895. Persoz hardness was performed according to ASTM D4366 method after coatings were cured at 23oC or 10oC and 50% RH for 1 day, 2 days, and 7 days. Shore D hardness was tested on a ¼ inch thick clear casting in a circular metal lid with diameter of 2.75 inches using 35 grams of materials in accordance with the method described in ASTM D2240. Gel time of a 150 grams mixture was recorded using Techne GT-3 Gelation Timer as the time after mixing the epoxy resin and curing agent to reach a defined point of viscosity. The gel timer is equipped with disposal plungers (22x5 mm) operated at one cycle per minute. Carbamation resistance was evaluated using the wet patch method described in ISO 2812. Coatings were applied onto a black Laneta card at 150 μm WFT using a bird bar. Following the specified cure time and temperature, coatings were exposed to a 1-inch square cotton patch saturated with water for 24 hours. Assessment of carbamation follows a relative scale of 1-5, as 5 being best with no signs of carbamation.
Concrete adhesion test was conducted in accordance with ASTM D7234 method. Epoxy coatings were applied to the surface of a concrete block at 10 mil thickness by a roller. The concrete block is 1.5-inch-thick, cast to ISO 13007-2 specification, and is sand blasted to have a surface profile of CSP 2 or 3 (International Concrete Repair Institute). The coatings were cured for minimum 7 days under specified conditions. Coatings were then drilled, and dollies were glued to the surface and the glue was cured overnight.
Dollies were pulled off using a portable pull-off adhesion tester. The adhesion strength and mode of failure were recorded.
Intercoat adhesion was evaluated by two methods. The first method is in accordance with ASTM D3359, Standard Test Methods for Measuring Adhesion by Tape Test (Test method A – X Cut Tape Test). The epoxy coating as primer was applied to a steel substrate, after specified time, a topcoat was applied on to the primer. The coatings were cured at 50% RH/23oC for 7 days before testing. The results were recorded as a relative scale of 1A-5A as 5A being best, no visible peeling or removal of coatings. The second method was conducted in accordance with ASTM D7234. The epoxy primer was coated at 10 mils thickness on a concrete substrate using a standard roller, followed by a topcoat at a specified time. The coatings were cured for 14 days and pull off adhesion test was performed.
Moisture vapor transmission testing was conducted in accordance with ASTM E96-13, Standard Test Methods for Water Vapor Transmission of Materials, at a set thickness in triplicate. The epoxy coatings were applied over a 1-inch thick concrete block by a roller and cured for 7 days before testing. The test specimens were fabricated for each coating thickness using the wet cup method with coating side facing 50% RH/23 oC and bottom side over water. Specimens were exposed over 6.75 X 10.75 X 2.0 inches stainless steel flanged pans using SM5143 vacuum sealant tape.
Near infrared spectroscopy samples were prepared by mixing about 5 grams of the curing agent-epoxy mixture using FlackTeK DAC 250 SP SpeedMixer™ by Hauschild. After mixing, a small amount of sample was placed in a disposable sample cell of 0.8 mm path length and placed in an oven at 25 oC. The near-infrared spectrometer Model 6500 was equipped with an Interactance probe by Foss NIR Systems, Inc. The IR spectra were collected over about 24 hours, and the spectra were analyzed using GRAMS software.
Degree of cure was determined by differential scanning calorimetry (DSC). About 5~10 mg of epoxy-curing agent mixture sample was analyzed by TA Instruments Q2000 DSC calibrated in T4P mode with Indium at a heating rate of 10 oC/minute. The sample was heated from -50 oC to 250 oC at 10 oC/minute, cooled back to -50C and the test was repeated. The degree of cure was determined by subtracting the residual heat of cure after specified cure time from the initial total heat of cure, divided by the initial total heat of cure.
Dynamic mechanical analysis (DMA) was performed on an RSA G2 rheometer (TA Instruments) outfitted with a thin film geometry. The formulations were prepared using two one-minute cycles on a FlackTek Speedmixer. The freshly prepared mixtures were cast at 125 um thickness and allowed to cure at ambient temperature and humidity for a minimum of 10 days. Dynamic mechanical data were then collected every 6°C over the -100 to 200 oC range using a one-minute soak time and a 6.28 rad/s deformation frequency. After the initial scan was collected, the same specimen was then retested (after being removed from the instrument and re-measuring its width and thickness). The midpoint glass transition temperature was measured as the peak in the tan delta curve.
Mechanical properties of epoxy castings in compressive mode were determined using a floor mounted dual column material testing machine, Instron 5582, equipped with 100 kN load cell and LVDT deflector sensor. Clear castings test samples were prepared with diluted bisphenol A/F resin to rectangular bars with 1-inch x 1-inch area for 7 days at ambient temperature. The bar was cut into 1-inch cubes and the test was conducted in accordance with ASTM D695 method at 0.1 inches/minute cross-head test speed using Bluehill 3 Universal Testing Software. Test was stopped at yield point, fracture or neither, then 25% deflection. Sample was placed between compression anvils (one attached to the baseplate of the Instron, the other attached to a 100 kN load cell.
Results and Discussion
Plasticizer Facilitates Cure in Epoxy Systems
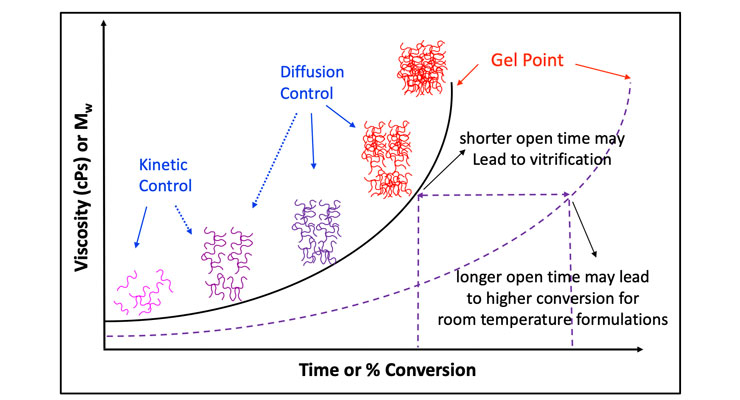
For two-component epoxy systems, the cure mechanism is driven by both reaction kinetics and diffusion process. As illustrated in Figure 1, in the early stage of cure, the dominant driving force is reaction kinetics, while at the latter stage, the cure is driven more by the diffusion process. The combination of both kinetics and diffusion rates determines the final properties of the cured system. Longer open time allows the resin and curing agent more time to react in the kinetics control region and reach higher conversion before gel point. Reaching the gel point too early can lead to molecular gridlock before all reactive groups can react and as a result poor conversion. Vitrification or B stage is a term used to describe such condition. Further reaction is extremely slow, and the cured system is typically brittle and demonstrates little toughness. There are several approaches to increase the molecular mobility and to extend the open time, for example, one common practice in the industry is adding non-reactive plasticizer such as benzyl alcohol to dilute the concentration of reacting species.
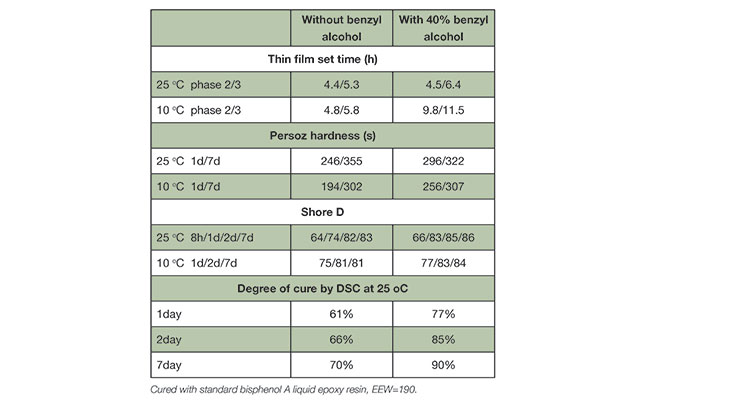
Table 1 summarizes the cured properties of isophorone diamine (IPD) with and without benzyl alcohol. Samples were cured with standard bisphenol A epoxy resin at 1:1 stoichiometry and cured at designated temperature and time period. For thin film set time, the clear coating without benzyl alcohol dried faster than that with 40% benzyl alcohol, both at ambient temperature and low temperature of 10 oC. This is consistent with the illustration in Figure 1 that plasticizer benzyl alcohol offers longer open time. Both systems showed similar Persoz pendulum hardness and shore D development however have much different physical property. Figure 2 shows the clear castings of samples with and without benzyl alcohol used for shore D measurement. It illustrates the importance of plasticizer benzyl alcohol to the cured properties where. Without benzyl alcohol, the specimen did not have physical strength, and was very brittle as indicated by cracks during shore D test and when they were dropped to floor, while the sample with benzyl alcohol had much higher toughness.
Property development has direct correlation to reaction conversion, which can be translated to the degree of epoxy cure. As shown in Figure 1, the presence of benzyl alcohol promotes higher conversion and higher degree of cure. The degree of cure was determined by differential scanning calorimetry (DSC) at 1 day, 2 days, and 7 days cure schedule.6 The presence of benzyl alcohol consistently increased degree of cure, and the system reached 90% cure after 7 days at ambient temperature in contrast to only 70% without benzyl alcohol.
Plasticizers like benzyl alcohol provide multiple benefits in terms of viscosity reduction, increased open time, and improved toughness of the cured system. On the other hand, plasticizer can potentially contribute to slow emission to the building inhabitants and environment throughout the service life of a floor. A challenge for the epoxy flooring technology is to reduce or eliminate the plasticizer in the formulation while maintaining high conversion and excellent physical properties.
New Curing Agents Deliver Excellent Cure Speed and Maintain High Conversion with Minimum to No Plasticizer
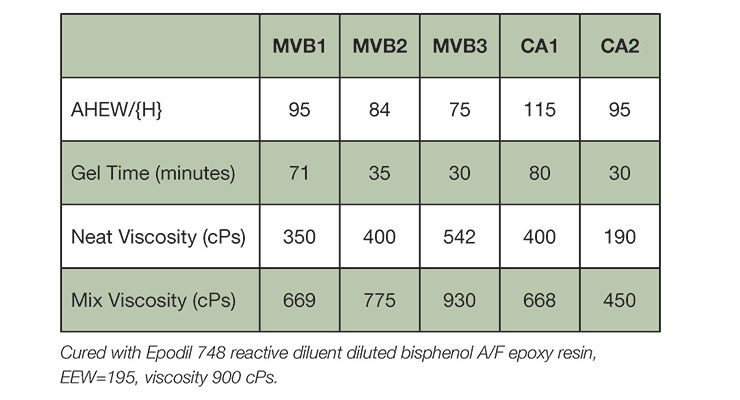
New curing agents MVB1, MVB2, and MVB3 were designed with minimum to no benzyl alcohol as plasticizer. They were evaluated against conventional benzyl alcohol containing cycloaliphatic curing agents CA1 and CA2. Both CA1 and CA2 are industrial standard cycloaliphatic curing agents that deliver good curing speed, aesthetics, mechanical property, and chemical resistance. CA1 is used for ambient temperature conditions, while CA2 offers low temperature cure down to 10°C with good working time, and cure speed. Table 2 summarizes the basic handling properties of the curing agents evaluated in this paper. As shown in Table 2, MVB1 has comparable gel time to CA1, and MVB2 and MVB3 to CA2.
Fast Property Development
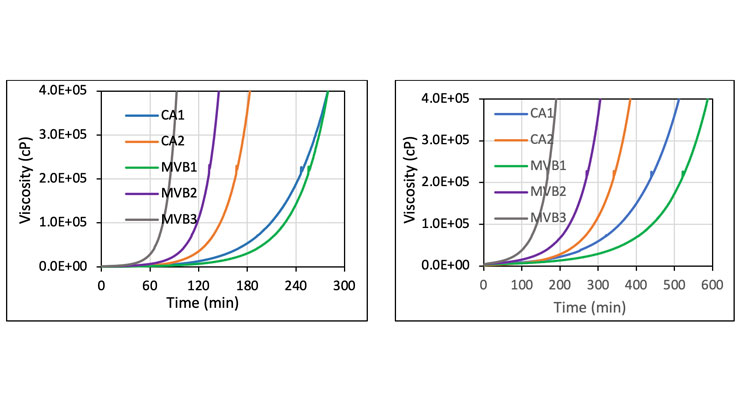
The new curing agents deliver fast cure speed and property development as demonstrated by cure viscosity build-up, and cure speed. Figure 3 shows the comparison of cure viscosity profiles of all curing agents at 25 oC and 10 oC. MVB3 exhibited fastest viscosity build-up at ambient and low temperature, followed by MVB2. CA1 showed similar viscosity increase to MVB1 at 25 oC, but faster increase at 10 oC. Fast viscosity build-up is often considered as the first indication of high reactivity. Table 3 summarizes the cure speed and hardness development. Clear coatings based on new curing agents MVB1, MVB2 and MVB3 curing agents provide fast cure at both ambient and low temperature conditions (10 oC). MVB1 showed cure speed measured by thin film set time between CA1 and CA2 at both ambient and 10 oC, while MVB2 and NVB3 offer much faster cure speed even at low temperature of 10 oC. At 10 oC, MVB1 displayed the slowest viscosity increase, which provided longer working time, while maintaining faster curing speed than CA1. In addition, the fast cure speed of MVB1, MVB2, and MVB3 also resulted in rapid mechanical property build in both coatings and thick castings. This is illustrated by the rapid early Persoz pendulum hardness development and Shore D build compared to CA1 and CA2. Typically, a minimal shore D of 50 is required for early “walk-on” for flooring application. The slower MVB1 based epoxy systems meet this condition between 1- and 2-days cure at 10 oC condition, while MVB2 and MVB3 systems deliver early “walk-on” in less than 24 hours at 10 oC. The range of cure speed offered by MVB1, MVB2, and MVB3 provides formulators versatility to choose from a faster or slower system to meet their specific requirement.
To better understand the phenomenon of viscosity build up and cure speed, in-depth fundamentals studies were conducted to investigate the cure mechanism by monitoring the curing process using analytical techniques such as near infrared spectroscopy, differential scanning calorimetry (DSC), and dynamic mechanical analysis (DMA).
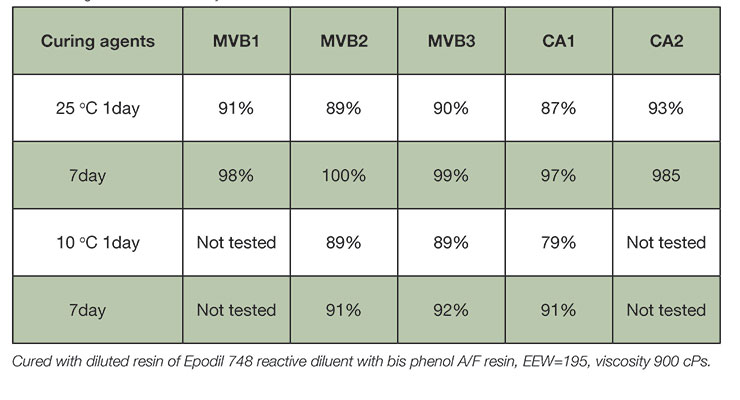
As shown in previous section, most epoxy systems require plasticizer like benzyl alcohol to reach high conversion and full property development. The new curing agents were developed with minimum to no benzyl alcohol and showed fast property development. This design principle ensures high degree of cure at ambient and sub-ambient temperature conditions, comparable to the benzyl alcohol containing curing agents CA1 and CA2. Table 4 displays the degree of cure as determined by DSC of resin-curing agent mixtures cured at ambient temperature and low temperature. Even at 10°C, MVB2 and MVB3 reach over 90% degree of cure after 7 days. High degree of cure is a feature particularly useful in industrial flooring application where ultimate performance depends on the development of mechanical, chemical and thermal resistance properties.
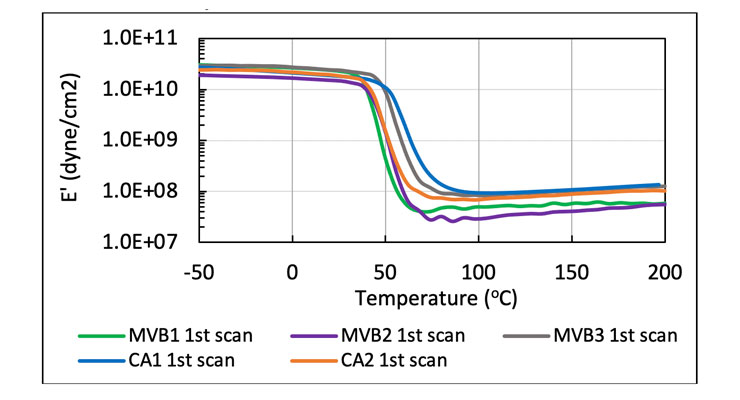
The high degree of cure is further corroborated by the dynamic mechanical analysis (DMA) results. DMA provides the mechanical property information such as storage modulus of the cured sample. Storage modulus (E') is a measure of elastic response of a viscoelastic material. It measures the stored energy.7 In this experiment, the curing agents were reacted with bisphenol A/F resin diluted with reactive diluent at 1:1 stoichiometry, and cured at ambient temperature and humidity for a week. Figure 4 shows the comparison of storage modulus, E’, of all the samples. E’ of all samples remained relatively flat within the test temperature to 200 oC, indicative of high degree of cure after 7 days, and no post cure in the rheometer with temperature ramp. It is typical for benzyl alcohol containing formulations such as CA1 and CA2 reaches high degree of cure. The unique chemistry in MVB1, MVB2 and MVB3 facilitates similar high degree of cure with minimum to no benzyl alcohol.
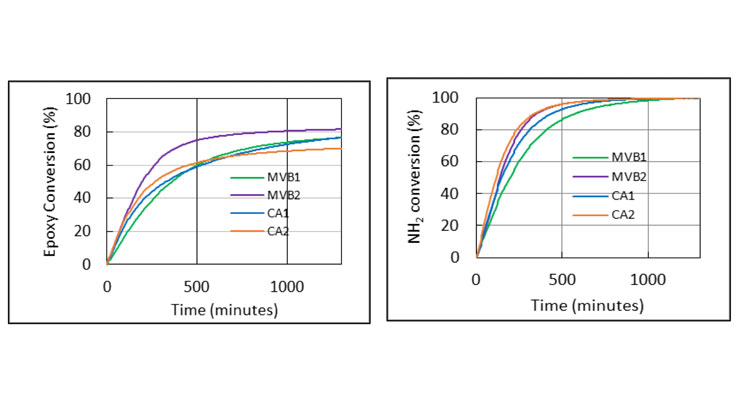
The DSC and DMA results show that the DSC method provides the overall rate of reaction based on the assumption that the rate of the heat release is proportional to the rate of reaction, and the DMA method indirectly points to whether a system has reached high degree of cure. However, no information on individual species or functional groups can be obtained using DSC or DMA. Fourier transform infrared spectroscopy (FTIR) spectroscopy not only provides information on the individual species of interest such as epoxy and amine groups, but also monitors the cure reactions in real time. The functional groups of interest for epoxide cure reactions typically have well-isolated absorption bands in the near-infrared region of the spectrum (1000 - 2500 nm). These factors make near infrared FTIR an attractive analytical tool to study the kinetics and mechanisms of epoxy resin cure reactions. As such, to delineate the reaction of epoxy and amine functional groups, near infrared FTIR was utilized to monitor the cure reaction and to semi-quantitatively determine the conversion of amine and epoxy groups.8 The conversion of oxirane (epoxy) and primary amine during the cure was monitored by the C-H stretch of oxirane ring at 1646 nm, and the N-H stretch of the primary amine at 2026 nm, respectively. Figure 5 illustrates the conversion of epoxy and primary amine during cure process. MVB2 has the fastest conversion of both epoxy group and primary amine group which correlates to the fastest cure speed. Primary amine groups readily react with epoxy group and generate secondary amines which react further with epoxy to form tertiary amine. As shown in Figure 5, for all curing agents tested, primary amine group reacted with epoxy to reach full conversion after 24 hours. Compared to CA1, MVB1 has similar rate and conversion of epoxy group after 24 hours, slower primary amine conversion early on and reaches the same complete consumption after 24 hours. This could correspond to the slower viscosity build up initially as shown in Figure 3. Although CA2 has fast conversion of primary amine groups, however, the percent conversion of epoxy is the lowest after 24 hours. MVB3 was not tested in this experiment, but we expect it to have high level of epoxy and primary amine conversion at a faster rate than MVB2.
New Curing Agents Based Coatings Offer Excellent Mechanical Properties and Improved Temperature Resistance
High Mechanical Strength
High compressive strength is critical for epoxy systems used in industrial floorings in order to protect the concrete structure and avoid structural damages. After the floor reaches a through-cured stage and is taken back into service, long-term mechanical integrity is paramount to ensure a long service life. Epoxy flooring materials are well known for their high mechanical property and it is important to understand if and how minimizing plasticizers will affect the mechanical properties of the flooring system. Evaluation of mechanical strength in compressive mode is a logical choice considering that the curing agents of interest are used for their high load bearing properties.
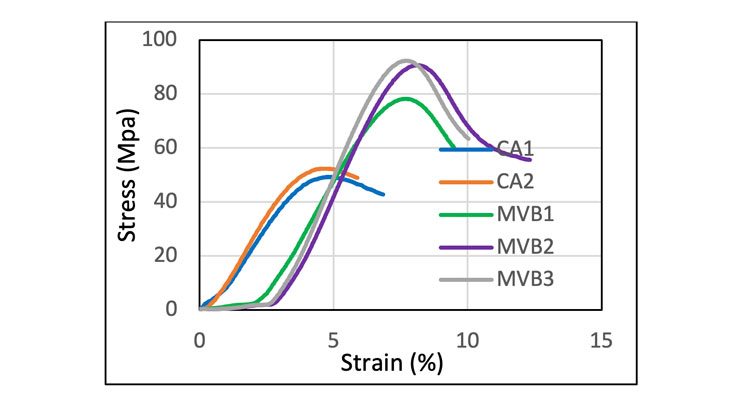
Compressive strength was determined in accordance with methods described in ASTM D635. Figure 6 shows the stress-strain curves of clear castings. Castings based on CA1 and CA2 show very similar results. Both provide about 50 MPa compressive strength at yield point with comparable elasticity in the linear region, 1.1 and 1.3 GPa respectively. The graph of MVB1, MVB2, and MVB3 based castings shows a longer toe followed by a linear region up to the yield point. In addition to the high yield strength of 78 for MVB1 and ~ 90 MPa for MVB2 and MVB3, all three MVB castings also display a much higher stiffness in the elastic region of ~2.2 GPa modulus, almost double of the modulus of CA1 and CA2, and much higher percentage strain. The higher modulus is a logical consequence of minimizing the plasticizers, since the plasticizers reduce the polymer crosslink density of the cured thermoset. Minimizing plasticisers in MVB1, MVB2, and MVB3 has also enhances mechanical integrity and offers a route to floorings with even higher mechanical strength and resilience for improved concrete protection.
Improved Temperature Resistance
Another potential drawback related to the use of plasticizers in amine-cured epoxy thermoset systems is the limited high temperature resistance. In conventional cycloaliphatic amine-based epoxies, plasticizers are essential to enable cure conversion at lower temperature conditions. Typically, an amine-cured epoxy coating will achieve a maximum Tg of about 55oC when cured at 25oC. Similarly, glass transition temperatures Tg of about 30-40 oC are found when the same epoxy thermoset is cured at 10 oC, though some post-curing may occur when temperatures are increased again after initial cure. The same principles of Tg development also apply to epoxy thermoset systems without plasticisers. It is therefore motivating to better understand the impact of temperature on mechanical integrity comparing conventional and new curing agents with minimum to no plasticizer.
Figure 7 shows the storage modulus of cured clear coatings as a function of temperature by DMA. The first scan is represented by solid lines versus the second scan by solid line with markers. For CA1 and CA2 based coatings, the second scan curves shifted significantly to higher temperature. Therefore, the glass transition temperature Tg of coatings based on CA1 and CA2 demonstrated a large increase between the first and second scan. Both CA1 and CA2 have first scan Tg of about 57 oC which in the second scan increased to 117oC and 108oC respectively, almost double. This behavior is illustrative for conventional epoxy thermoset systems where the presence of non-reactive plasticizers can exit the coating upon exposure to high temperature. On the contrary, MVB1, MVB2, and MVB3 based coatings show no such shortfall. The increase in Tg between first and second scan is much smaller than that of CA1 and CA2, <15 oC for MVB1, MBV2, and MBV3. Figure 7 clearly demonstrates the improved temperature resistance property of coatings based on new curing agents. This temperature resistance feature is especially beneficial for flooring exposed to high temperatures, such as factories or kitchens, therefore prolongs the service life of a floor and provides cost-saving benefit to floor owner.
New Curing Agents Provide Excellent Adhesion to Concrete and Barrier Property to Moisture Vapor Transmission
In previous sections, we demonstrate the advantages of the new curing agents MVB1, MVB2, and MVB3 which allow coating formulators to design formulations with minimum to no plasticizer. These formulations offer tailorable cure speed, fast property development, and excellent mechanical properties. In this section, we will focus on the benefits of using these new curing agents as a moisture vapor barrier to address the moisture transmission issue in concrete.
Adhesion to Concrete
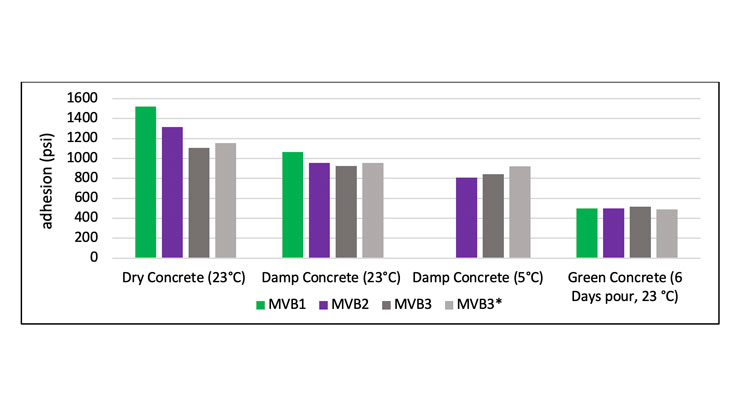
Adhesion to concrete is one of the key requirements for a moisture vapor barrier coating as it is the first line of defense against the moisture vapor transmission from the concrete slab. The moisture vapor barrier coatings are applied to concrete that can contain varying amount of water depending on the age and the condition of the concrete. The vapor barrier is required to adhere to both dry and damp concrete to avoid any failures of subsequent materials placed or coated on top. Dry and damp concrete in this study are defined as concrete cured in a controlled environment (50% RH/23 oC) greater than 28 days after casting, and green concrete as a fresh concrete cured for 6 days. All concrete blocks had a surface profile of CSP 2 or 3 (International Concrete Repair Institute) as would be recommended for surface preparation. For damp concrete testing, the concrete block was submerged in water for 24 hours, and then removed from water for testing. The relative moisture content was determined by a Testo 606-1 material moisture meter that was set to material number 3 (cement screed, concrete). Dry and damp concrete contains 1.5% and 3% of moisture respectively. The coatings were applied by roller at 10 mil wet film thickness and cured at 50% RH/23 oC for 7 days. Adhesion testing was conducted in accordance with ASTM D7234 method. Figure 7 displays the excellent adhesion of all moisture vapor barrier coatings to dry and damp 28 day cure concrete, and 6 day pour green concrete. Green concrete is an aggressive test as it contains a significant amount of moisture in the concrete and the properties of the concrete are not yet fully developed. Excellent adhesion of moisture vapor barrier coatings to concrete substrates provides a good foundation for the subsequent layer to be coated on.
Carbamation Resistance, Intercoat Adhesion, and Overcoatability
Intercoat adhesion is crucial for a moisture vapor barrier coating since it is a primer and serves as a tie coating between the concrete substrate and the subsequent coatings applied on top. One fundamental study to investigate whether a coating has good intercoat adhesion is a carbamation resistance test. Carbamation occurs when low molecular weight amines from a curing agent migrate from the bulk of the coating to the surface, where they react with carbon dioxide and moisture from the air to form ammonium salts. This process is highly undesirable as it competes with the needed amine-epoxy reaction. It is favored especially at lower temperature and high humidity and manifests itself as haziness, reduced gloss, or white salt on the surface. Carbamation not only reduces the visual appeal of the coating, but also leads to poor intercoat adhesion. Carbamation resistance is critical for a primer to ensure good intercoat adhesion to the subsequent layer coated on top.
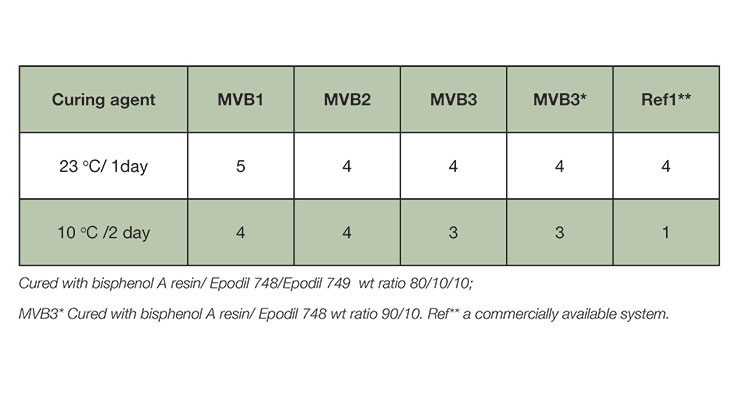
Carbamation resistance testing was evaluated using the wet patch method described in ISO 2812. Coatings were applied onto a black Laneta card and exposed to a cotton patch saturated with water for 24 hours following the specified cure time and temperature. Assessment of carbamation follows a relative scale 1-5, as 5 being best with no signs of carbamation. New curing agents were benchmarked against a commercially available fast curing agent used as moisture barrier, Ref1. Table 5 summarizes the carbamation test results at ambient and 10 oC. All new curing agents show good carbmation resistance at ambient temperature. Reference fast curing agent showed very poor result at low temperature of 10 oC.
Overcoatability is another important criterium for a moisture vapor barrier coating (MVB). Generally, most commercial floorings have a multi-layer system to get the highest performance. For a seamless flooring system, a primer coating is followed by a mid-coat and/or topcoat. The mid-coat and topcoat could be filled, pigmented or clear coatings and could be any of the vast thermoplastic or thermoset technologies including epoxies, polyurethanes, polyureas, and acrylics.
For a resilient flooring system, a primer coating is followed by a self-leveling coating such as a cementitious overlay or other thick build coating to smooth out the floor. The final step would be to apply a floor covering such as wood, tile, or carpet.
The overcoatability is determined by the intercoat adhesion of the moisture vapor barrier layer to the subsequent layer following a specified schedule. It also provides information on overcoat window.
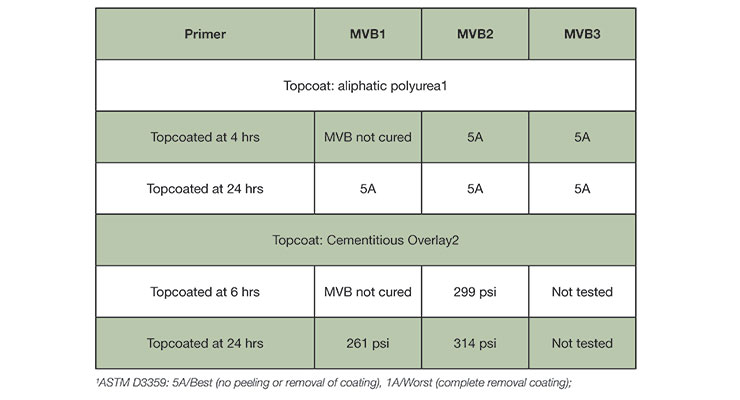
Two test methods were employed to assess the intercoat adhesion. In the first test, a commercial aliphatic polyurea was coated at 10 mil thickness on the surface of the 10 mil thick MVB1 or MVB2 coatings after a cure time of 4 hours and 24 hours cure at ambient conditions (50% RH/23oC). The intercoat adhesion test was conducted based on the method described in ASTM D3359 after the system was cured for 7 days cure at 50% RH/23 oC, and the results were reported on a scale of 1-5 with 5 being the best.
The data in Table 6 shows that all MVB coatings provide excellent intercoat adhesion to an aliphatic polyurea topcoat. For fast curing agents MVB2 and MVB3, the topcoat can be applied in 4 hours with excellent intercoat adhesion. This provides a solution for fast return to service where multiple layers can be applied within one day.
Excellent overcoatability is also demonstrated in a thick build cementitious overlay. In this test, a commercial cementitious overlay was applied on top of MVB1 and MVB2 at 1/8-inch thickness using a small trowel after a minimum time of 6 hours and 24 hours cure at 50% RH/23°C. The intercoat adhesion testing was conducted using ASTM D7234 after 14 days cure. The cementitious overlay also showed good intercoat adhesion to the new MVB layer (Table 6).
The overlay has a strong bond to the MVB coatings as the dollie pull leads to cohesive failure of the cementitious overlay instead of the adhesive failure at the interface. For both MVB1 and MVB2, the test was done within 24 hours. It is possible for the MVB coatings to have an overcoat window beyond 24 hours but this was not tested in this study. We also expect MVB3 would perform similarly to MVB2 based on the design principle of the curing agent.
Barrier to Moisture Vapor Transmission
Concrete is a permeable material that allows moisture underneath the concrete, or trapped in the concrete itself, to migrate as a vapor phase to the concrete surface. This moisture migration can cause floor failure and result in significant financial loss. Applying an effective moisture vapor barrier (MVB) on the surface of the concrete can significantly reduce moisture vapor from migrating out of the concrete slab due to hydrostatic pressure or capillary flow. The MVB should have a very low moisture vapor transmission or flow through the coating because even a small amount of moisture collected under the non-permeable floor covering can lead to adhesive failures, delamination, or microbial growth.
One of the most common techniques to measure moisture vapor transmission (MVT) of a coating is using ASTM E96 method. In this method, test specimens at each coating thickness are fabricated to the dimensions of a flanged stainless steel pans as specified in the method. The pans are filled partly with water, then the tested materials are tightly fitted to seal the pans and monitored at controlled environment (23°C/50% RH).
The test specimens are weighed regularly to the nearest 0.0001 gram and this process continues until enough data points are collected to establish a moisture vapor transmission rate through the coating. The permeance value is calculated from the moisture vapor transmission rate in perms (grain of water per hour per square feet per inch of mercury) and the results is recorded for each test specimen. ASTM F3010 certification requires a very low permeance value of 0.1 perm or lower to be an effective moisture
vapor barrier.
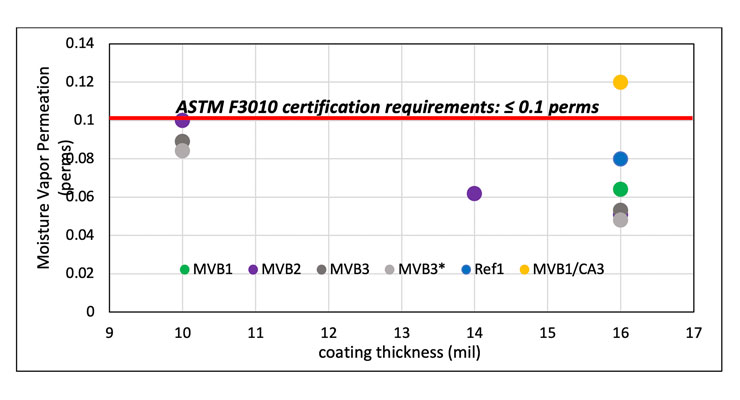
The moisture vapor transmission rate of the new curing agents, measured using ASTM E96 method, and benchmarked alongside a 50/50 blend of MVB1 and a benzyl alcohol containing curing agent CA39 and a commercially available fast cure MVB system Ref1. The permeance results are shown in Figure 8. All new MVB curing agents outperform Ref1 with lower permeance at the same thickness of 16 mil or lower thickness of 14 mil. MVB1, MVB2, and MVB3 show two times lower permeance than the MVB1/CA3 blend at the same coating thickness of 16 mils.
Even at very thin thickness of 10 mil, MVB2 and MVB3 display low water vapor transmission to meet the ASTM F3010 certification requirement of 0.1 perms. MVB1/CA3 blend contains about 20% benzyl alcohol and it is hypothesized that some benzyl alcohol could be emitting out of the coating over time leaving voids in the film. The voids could provide channels for moisture vapor to pass through.
Even at low thickness of 10 mils, all new MVB curing agents outperforms MVB1/CA3 at 16 mil. The moisture vapor testing indicates that both MVB1, MVB2, and MVB3 can be utilized as an effective moisture vapor barrier due to very low permeance to water vapor. And the high performance at lower thickness translates to cost-saving for end-users.
Conclusions
We have demonstrated in this paper epoxy systems using new amine curing agent technology that offers tailorable cure speed, fast property development and good carbamation resistance. The new technology enables the design of epoxy systems with minimum to no plasticizer. Conventional cycloaliphatic amine-cured epoxies without plasticizers can suffer from incomplete conversion and poor performance. The new amine curing agents presented in this paper, MVB1, MVB2 and MVB3, containing minimum to no plasticizer, deliver high conversion and better mechanical property. They provide an alternative to conventional plasticizer containing curing agents to meet compliance with stringent emission requirement.
The paper details fundamental study of new curing agents MVB1, MVB2 and MVB3, against conventional plasticizer containing curing agents CA1 and CA2 in flooring applications. Various analytical techniques were employed to understand the cure behavior of these curing agents. DSC, DMA and near infrared data corroborated with each other to confirm high reaction conversion of these new epoxy systems, comparable to conventional plasticizer containing system using CA1 and CA2. Minimizing or eliminating plasticizer in MVB1, MVB2 and MVB3 did not result in negative effects of low conversion and compromised mechanical properties. On the contrary, compressive testing showed MVB1, MVB2 and MVB3 provide coatings with higher compressive strength and toughness compared to CA1 and CA2. In addition, DMA test demonstrated minimizing plasticizer imparted better high temperature resistance.
Furthermore, MVB1, MVB2 and MVB3 provide outstanding adhesion to dry, damp and green concrete. All showed excellent intercoat adhesion to organic coatings such as polyurea, or thick cementitious overlayer. Mostly importantly, MVB1, MVB2 and MVB3 are proven to be excellent barriers to moisture vapor transmission at coating thickness of 10 mil and have been used as excellent moisture vapor barriers to address moisture vapor transmission in concrete.
The performance features demonstrated in this paper show that MVB1, MVB2 and MVB3 are great products for epoxy floor coatings to meet stringent emission requirement, more importantly they are excellent choice for moisture vapor barrier to meet ASTM F3010 certification of less than 0.1 perm even at 10 mil thickness.
Acknowledgements
The authors would like to thank Mike Oberlander, Dave Brown, and Edwin Lijffijt for their contributions in the experimental work presented in this paper.
References
1. “Moisture in Concrete and Moisture-sensitive Finishes and Coatings” in Cement Concrete & Aggregates Australia April 2007.
2. Tanaka, Y., “Synthesis and characteristics of epoxides”, in C.A. May, ed., “Epoxy Resins Chemistry and Technology” Marcel Dekker, 1988
3. Wissanrakkit, G. and Gilham, J.K., J. of Coatings Technology, 1990, 62 (783), 35-36.
4. Abbreviations of amine curing agents and further details are included in the respective technical datasheets from Evonik Corporation: Ancamine® 2739 curing agent (MVB1), Ancamine® 2800 curing agent (MVB2), Ancamine® 2850 curing agent (MVB3), Ancamine® 1618 curing agent (CA1), and Ancamine® 2519 curing agent (CA2).
5. Epodil® 748 Reactive Diluent, Evonik Corporation.
6. (a) Lee, W. I., Loos, A. C., Springer, G. S. J. Composite Mat. 1982, 16, 510. (b) Dangayach, K., Smith, C. J. Coatings Technology, 1996, 68 (863), 29-37.
7. Vratsanos, M. Coatings Tech July 2017, 28-38.
8. (a) Xu, L.; Fu, J. H.; Schlup, J. R. J. Am. Chem. Soc. 1994, 116, 2821-2826; (b) Fu, J. H.; Schlup, J. R. J. Appl. Polymer Sci. 1993, 49, 219-227.
9. Ancamine® 2764 curing agent (CA3).
Two-component epoxy technology provides excellent chemical resistance, mechanical strength and adhesion to various substrates. It has been widely used in many different applications, especially in concrete coatings. Traditionally, many epoxy systems require the use of plasticizer to ensure full chemical conversion of the polymer matrix. One of the challenges from epoxy floor coating application is the potential emission of plasticizer to the building inhabitants and environment throughout the service life of a floor. Legislation and end-user demand have driven new product development to focus on mitigating potential emissions from the plasticizer, as well as maintaining high performance.
Epoxy concrete coatings are essential to improve aesthetics, durability and provide protection to concrete substrates. Another challenge for concrete floor coatings is the moisture vapor transmission through the concrete floor. Moisture can cause deterioration of flooring adhesives and delamination of coatings by osmotic action, thus reducing or eliminating the benefits of epoxy coatings on concrete. It is one of the frequent causes for floor failure. The cost to replace a failed floor can be significant. Recent innovative technology developments have offered a solution to remediate the issue of moisture vapor transmission through the concrete floor.
This paper will present epoxy systems based on innovative curing agent technology that contains very low to no plasticizer and has excellent moisture vapor barrier property as thin coating. Fundamental study highlights the full property development of these systems. The paper will discuss the key performance attributes using these new curing agent systems. A combination of high barrier property to moisture vapor, low to no plasticizer, tailorable working time, and fast property development offers coating formulators the choices to design new floor systems incorporating thin epoxy coatings as effective moisture vapor barrier to meet specific requirements.
Introduction
Concrete is one of the most used construction materials due to its strength, durability, resilience, safety, and low cost. In flooring applications, demand for concrete for both interior and exterior use in residential and commercial settings is growing significantly thanks to the strong construction market around the globe. This growth includes new housing jobs as well as refurbishing and remodeling work. Concrete coatings have multiple functions where they can serve as an impermeable barrier, protect concrete from wear, deterioration, and contamination, enhance physical performance, provide chemical resistance, and improve aesthetics. An impermeable barrier is essential for concrete floorings. Since concrete is a permeable material, it allows not only chemicals from above to permeate and attack the concrete, but also the moisture originating from below the concrete or trapped in the concrete itself, to migrate as a vapor phase to concrete surface. Cured concrete traps varied amount of moisture, about 1%-2% in ambient dry concrete, and 4% to 5% in wet concrete.1
Moisture transfer phenomenon accounts for many flooring failures resulting in significant financial losses annually. Excessive moisture leads to damaged floor coverings, safety hazards due to slips and falls, and microbial growth leading to reduced indoor air quality. A moisture vapor barrier coating is proven to be an effective solution to address flooring failures by minimizing moisture vapor transmission. Applicators can apply a moisture vapor barrier coating for both existing and new construction jobs before applying the resilient floor covering, such as tile, wood, carpet, laminate, or installing a seamless flooring system of multiple coating layers for example broadcast floors, or Terrazzo.
Well-designed epoxy floor coatings are often used as moisture vapor barrier due to their excellent barrier property. Many epoxy systems require the use of reactive epoxy diluent, or plasticizer such as benzyl alcohol to ensure full chemical conversion of the polymer matrix by reducing the concentration of reactive groups and increasing mobility of the reactive species.2 These approaches enable a sufficient degree of cure well beyond the gel point to provide high performance of the flooring product.3 One of the challenges from epoxy floor coatings application is the potential emission of plasticizer to the building inhabitants and environment throughout the service life of a floor. One area of the new product development for epoxy floor coatings is to minimize the potential emissions from the plasticizer.
The primary objective of this paper is to present the development of new curing agent technology that contains very low to no plasticizer content and has excellent moisture vapor barrier properties as thin coating. The paper will include the comparison study of the new technology versus the conventional plasticizer containing products and the key performance benefits using the new curing agents. The new curing agent systems display a high degree of cure at ambient and low temperature, excellent moisture vapor barrier properties, and improved resistance to mechanical stress and temperature exposures.
Experimental
The new amine curing agents used in this study as moisture vapour barrier are represented as MVB1, MVB2, and MVB3, and the benchmarking conventional curing agents as CA1 and CA2.4 All coatings or castings were prepared with standard bisphenol A liquid epoxy resin or bisphenol A/F epoxy resin diluted with a glycidyl ether of C12-14 alcohol at 1:1 stoichiometry unless otherwise specified.5
The viscosity profiles were obtained on a Brookfield viscometer at 25oC using about 12 grams of mixed material. Coatings for thin film set time and Persoz hardness were deposited on glass substrates at 150 µm wet film thickness (WFT). The thin film set time (TFST) was determined using a Beck-Koller recorder, in accordance with ASTM D5895. Persoz hardness was performed according to ASTM D4366 method after coatings were cured at 23oC or 10oC and 50% RH for 1 day, 2 days, and 7 days. Shore D hardness was tested on a ¼ inch thick clear casting in a circular metal lid with diameter of 2.75 inches using 35 grams of materials in accordance with the method described in ASTM D2240. Gel time of a 150 grams mixture was recorded using Techne GT-3 Gelation Timer as the time after mixing the epoxy resin and curing agent to reach a defined point of viscosity. The gel timer is equipped with disposal plungers (22x5 mm) operated at one cycle per minute. Carbamation resistance was evaluated using the wet patch method described in ISO 2812. Coatings were applied onto a black Laneta card at 150 μm WFT using a bird bar. Following the specified cure time and temperature, coatings were exposed to a 1-inch square cotton patch saturated with water for 24 hours. Assessment of carbamation follows a relative scale of 1-5, as 5 being best with no signs of carbamation.
Concrete adhesion test was conducted in accordance with ASTM D7234 method. Epoxy coatings were applied to the surface of a concrete block at 10 mil thickness by a roller. The concrete block is 1.5-inch-thick, cast to ISO 13007-2 specification, and is sand blasted to have a surface profile of CSP 2 or 3 (International Concrete Repair Institute). The coatings were cured for minimum 7 days under specified conditions. Coatings were then drilled, and dollies were glued to the surface and the glue was cured overnight.
Dollies were pulled off using a portable pull-off adhesion tester. The adhesion strength and mode of failure were recorded.
Intercoat adhesion was evaluated by two methods. The first method is in accordance with ASTM D3359, Standard Test Methods for Measuring Adhesion by Tape Test (Test method A – X Cut Tape Test). The epoxy coating as primer was applied to a steel substrate, after specified time, a topcoat was applied on to the primer. The coatings were cured at 50% RH/23oC for 7 days before testing. The results were recorded as a relative scale of 1A-5A as 5A being best, no visible peeling or removal of coatings. The second method was conducted in accordance with ASTM D7234. The epoxy primer was coated at 10 mils thickness on a concrete substrate using a standard roller, followed by a topcoat at a specified time. The coatings were cured for 14 days and pull off adhesion test was performed.
Moisture vapor transmission testing was conducted in accordance with ASTM E96-13, Standard Test Methods for Water Vapor Transmission of Materials, at a set thickness in triplicate. The epoxy coatings were applied over a 1-inch thick concrete block by a roller and cured for 7 days before testing. The test specimens were fabricated for each coating thickness using the wet cup method with coating side facing 50% RH/23 oC and bottom side over water. Specimens were exposed over 6.75 X 10.75 X 2.0 inches stainless steel flanged pans using SM5143 vacuum sealant tape.
Near infrared spectroscopy samples were prepared by mixing about 5 grams of the curing agent-epoxy mixture using FlackTeK DAC 250 SP SpeedMixer™ by Hauschild. After mixing, a small amount of sample was placed in a disposable sample cell of 0.8 mm path length and placed in an oven at 25 oC. The near-infrared spectrometer Model 6500 was equipped with an Interactance probe by Foss NIR Systems, Inc. The IR spectra were collected over about 24 hours, and the spectra were analyzed using GRAMS software.
Degree of cure was determined by differential scanning calorimetry (DSC). About 5~10 mg of epoxy-curing agent mixture sample was analyzed by TA Instruments Q2000 DSC calibrated in T4P mode with Indium at a heating rate of 10 oC/minute. The sample was heated from -50 oC to 250 oC at 10 oC/minute, cooled back to -50C and the test was repeated. The degree of cure was determined by subtracting the residual heat of cure after specified cure time from the initial total heat of cure, divided by the initial total heat of cure.
Dynamic mechanical analysis (DMA) was performed on an RSA G2 rheometer (TA Instruments) outfitted with a thin film geometry. The formulations were prepared using two one-minute cycles on a FlackTek Speedmixer. The freshly prepared mixtures were cast at 125 um thickness and allowed to cure at ambient temperature and humidity for a minimum of 10 days. Dynamic mechanical data were then collected every 6°C over the -100 to 200 oC range using a one-minute soak time and a 6.28 rad/s deformation frequency. After the initial scan was collected, the same specimen was then retested (after being removed from the instrument and re-measuring its width and thickness). The midpoint glass transition temperature was measured as the peak in the tan delta curve.
Mechanical properties of epoxy castings in compressive mode were determined using a floor mounted dual column material testing machine, Instron 5582, equipped with 100 kN load cell and LVDT deflector sensor. Clear castings test samples were prepared with diluted bisphenol A/F resin to rectangular bars with 1-inch x 1-inch area for 7 days at ambient temperature. The bar was cut into 1-inch cubes and the test was conducted in accordance with ASTM D695 method at 0.1 inches/minute cross-head test speed using Bluehill 3 Universal Testing Software. Test was stopped at yield point, fracture or neither, then 25% deflection. Sample was placed between compression anvils (one attached to the baseplate of the Instron, the other attached to a 100 kN load cell.
Results and Discussion
Plasticizer Facilitates Cure in Epoxy Systems
For two-component epoxy systems, the cure mechanism is driven by both reaction kinetics and diffusion process. As illustrated in Figure 1, in the early stage of cure, the dominant driving force is reaction kinetics, while at the latter stage, the cure is driven more by the diffusion process. The combination of both kinetics and diffusion rates determines the final properties of the cured system. Longer open time allows the resin and curing agent more time to react in the kinetics control region and reach higher conversion before gel point. Reaching the gel point too early can lead to molecular gridlock before all reactive groups can react and as a result poor conversion. Vitrification or B stage is a term used to describe such condition. Further reaction is extremely slow, and the cured system is typically brittle and demonstrates little toughness. There are several approaches to increase the molecular mobility and to extend the open time, for example, one common practice in the industry is adding non-reactive plasticizer such as benzyl alcohol to dilute the concentration of reacting species.
Table 1 summarizes the cured properties of isophorone diamine (IPD) with and without benzyl alcohol. Samples were cured with standard bisphenol A epoxy resin at 1:1 stoichiometry and cured at designated temperature and time period. For thin film set time, the clear coating without benzyl alcohol dried faster than that with 40% benzyl alcohol, both at ambient temperature and low temperature of 10 oC. This is consistent with the illustration in Figure 1 that plasticizer benzyl alcohol offers longer open time. Both systems showed similar Persoz pendulum hardness and shore D development however have much different physical property. Figure 2 shows the clear castings of samples with and without benzyl alcohol used for shore D measurement. It illustrates the importance of plasticizer benzyl alcohol to the cured properties where. Without benzyl alcohol, the specimen did not have physical strength, and was very brittle as indicated by cracks during shore D test and when they were dropped to floor, while the sample with benzyl alcohol had much higher toughness.
Property development has direct correlation to reaction conversion, which can be translated to the degree of epoxy cure. As shown in Figure 1, the presence of benzyl alcohol promotes higher conversion and higher degree of cure. The degree of cure was determined by differential scanning calorimetry (DSC) at 1 day, 2 days, and 7 days cure schedule.6 The presence of benzyl alcohol consistently increased degree of cure, and the system reached 90% cure after 7 days at ambient temperature in contrast to only 70% without benzyl alcohol.
Plasticizers like benzyl alcohol provide multiple benefits in terms of viscosity reduction, increased open time, and improved toughness of the cured system. On the other hand, plasticizer can potentially contribute to slow emission to the building inhabitants and environment throughout the service life of a floor. A challenge for the epoxy flooring technology is to reduce or eliminate the plasticizer in the formulation while maintaining high conversion and excellent physical properties.
New Curing Agents Deliver Excellent Cure Speed and Maintain High Conversion with Minimum to No Plasticizer
New curing agents MVB1, MVB2, and MVB3 were designed with minimum to no benzyl alcohol as plasticizer. They were evaluated against conventional benzyl alcohol containing cycloaliphatic curing agents CA1 and CA2. Both CA1 and CA2 are industrial standard cycloaliphatic curing agents that deliver good curing speed, aesthetics, mechanical property, and chemical resistance. CA1 is used for ambient temperature conditions, while CA2 offers low temperature cure down to 10°C with good working time, and cure speed. Table 2 summarizes the basic handling properties of the curing agents evaluated in this paper. As shown in Table 2, MVB1 has comparable gel time to CA1, and MVB2 and MVB3 to CA2.
Fast Property Development
The new curing agents deliver fast cure speed and property development as demonstrated by cure viscosity build-up, and cure speed. Figure 3 shows the comparison of cure viscosity profiles of all curing agents at 25 oC and 10 oC. MVB3 exhibited fastest viscosity build-up at ambient and low temperature, followed by MVB2. CA1 showed similar viscosity increase to MVB1 at 25 oC, but faster increase at 10 oC. Fast viscosity build-up is often considered as the first indication of high reactivity. Table 3 summarizes the cure speed and hardness development. Clear coatings based on new curing agents MVB1, MVB2 and MVB3 curing agents provide fast cure at both ambient and low temperature conditions (10 oC). MVB1 showed cure speed measured by thin film set time between CA1 and CA2 at both ambient and 10 oC, while MVB2 and NVB3 offer much faster cure speed even at low temperature of 10 oC. At 10 oC, MVB1 displayed the slowest viscosity increase, which provided longer working time, while maintaining faster curing speed than CA1. In addition, the fast cure speed of MVB1, MVB2, and MVB3 also resulted in rapid mechanical property build in both coatings and thick castings. This is illustrated by the rapid early Persoz pendulum hardness development and Shore D build compared to CA1 and CA2. Typically, a minimal shore D of 50 is required for early “walk-on” for flooring application. The slower MVB1 based epoxy systems meet this condition between 1- and 2-days cure at 10 oC condition, while MVB2 and MVB3 systems deliver early “walk-on” in less than 24 hours at 10 oC. The range of cure speed offered by MVB1, MVB2, and MVB3 provides formulators versatility to choose from a faster or slower system to meet their specific requirement.
To better understand the phenomenon of viscosity build up and cure speed, in-depth fundamentals studies were conducted to investigate the cure mechanism by monitoring the curing process using analytical techniques such as near infrared spectroscopy, differential scanning calorimetry (DSC), and dynamic mechanical analysis (DMA).
As shown in previous section, most epoxy systems require plasticizer like benzyl alcohol to reach high conversion and full property development. The new curing agents were developed with minimum to no benzyl alcohol and showed fast property development. This design principle ensures high degree of cure at ambient and sub-ambient temperature conditions, comparable to the benzyl alcohol containing curing agents CA1 and CA2. Table 4 displays the degree of cure as determined by DSC of resin-curing agent mixtures cured at ambient temperature and low temperature. Even at 10°C, MVB2 and MVB3 reach over 90% degree of cure after 7 days. High degree of cure is a feature particularly useful in industrial flooring application where ultimate performance depends on the development of mechanical, chemical and thermal resistance properties.
The high degree of cure is further corroborated by the dynamic mechanical analysis (DMA) results. DMA provides the mechanical property information such as storage modulus of the cured sample. Storage modulus (E') is a measure of elastic response of a viscoelastic material. It measures the stored energy.7 In this experiment, the curing agents were reacted with bisphenol A/F resin diluted with reactive diluent at 1:1 stoichiometry, and cured at ambient temperature and humidity for a week. Figure 4 shows the comparison of storage modulus, E’, of all the samples. E’ of all samples remained relatively flat within the test temperature to 200 oC, indicative of high degree of cure after 7 days, and no post cure in the rheometer with temperature ramp. It is typical for benzyl alcohol containing formulations such as CA1 and CA2 reaches high degree of cure. The unique chemistry in MVB1, MVB2 and MVB3 facilitates similar high degree of cure with minimum to no benzyl alcohol.
The DSC and DMA results show that the DSC method provides the overall rate of reaction based on the assumption that the rate of the heat release is proportional to the rate of reaction, and the DMA method indirectly points to whether a system has reached high degree of cure. However, no information on individual species or functional groups can be obtained using DSC or DMA. Fourier transform infrared spectroscopy (FTIR) spectroscopy not only provides information on the individual species of interest such as epoxy and amine groups, but also monitors the cure reactions in real time. The functional groups of interest for epoxide cure reactions typically have well-isolated absorption bands in the near-infrared region of the spectrum (1000 - 2500 nm). These factors make near infrared FTIR an attractive analytical tool to study the kinetics and mechanisms of epoxy resin cure reactions. As such, to delineate the reaction of epoxy and amine functional groups, near infrared FTIR was utilized to monitor the cure reaction and to semi-quantitatively determine the conversion of amine and epoxy groups.8 The conversion of oxirane (epoxy) and primary amine during the cure was monitored by the C-H stretch of oxirane ring at 1646 nm, and the N-H stretch of the primary amine at 2026 nm, respectively. Figure 5 illustrates the conversion of epoxy and primary amine during cure process. MVB2 has the fastest conversion of both epoxy group and primary amine group which correlates to the fastest cure speed. Primary amine groups readily react with epoxy group and generate secondary amines which react further with epoxy to form tertiary amine. As shown in Figure 5, for all curing agents tested, primary amine group reacted with epoxy to reach full conversion after 24 hours. Compared to CA1, MVB1 has similar rate and conversion of epoxy group after 24 hours, slower primary amine conversion early on and reaches the same complete consumption after 24 hours. This could correspond to the slower viscosity build up initially as shown in Figure 3. Although CA2 has fast conversion of primary amine groups, however, the percent conversion of epoxy is the lowest after 24 hours. MVB3 was not tested in this experiment, but we expect it to have high level of epoxy and primary amine conversion at a faster rate than MVB2.
New Curing Agents Based Coatings Offer Excellent Mechanical Properties and Improved Temperature Resistance
High Mechanical Strength
High compressive strength is critical for epoxy systems used in industrial floorings in order to protect the concrete structure and avoid structural damages. After the floor reaches a through-cured stage and is taken back into service, long-term mechanical integrity is paramount to ensure a long service life. Epoxy flooring materials are well known for their high mechanical property and it is important to understand if and how minimizing plasticizers will affect the mechanical properties of the flooring system. Evaluation of mechanical strength in compressive mode is a logical choice considering that the curing agents of interest are used for their high load bearing properties.
Compressive strength was determined in accordance with methods described in ASTM D635. Figure 6 shows the stress-strain curves of clear castings. Castings based on CA1 and CA2 show very similar results. Both provide about 50 MPa compressive strength at yield point with comparable elasticity in the linear region, 1.1 and 1.3 GPa respectively. The graph of MVB1, MVB2, and MVB3 based castings shows a longer toe followed by a linear region up to the yield point. In addition to the high yield strength of 78 for MVB1 and ~ 90 MPa for MVB2 and MVB3, all three MVB castings also display a much higher stiffness in the elastic region of ~2.2 GPa modulus, almost double of the modulus of CA1 and CA2, and much higher percentage strain. The higher modulus is a logical consequence of minimizing the plasticizers, since the plasticizers reduce the polymer crosslink density of the cured thermoset. Minimizing plasticisers in MVB1, MVB2, and MVB3 has also enhances mechanical integrity and offers a route to floorings with even higher mechanical strength and resilience for improved concrete protection.
Improved Temperature Resistance
Another potential drawback related to the use of plasticizers in amine-cured epoxy thermoset systems is the limited high temperature resistance. In conventional cycloaliphatic amine-based epoxies, plasticizers are essential to enable cure conversion at lower temperature conditions. Typically, an amine-cured epoxy coating will achieve a maximum Tg of about 55oC when cured at 25oC. Similarly, glass transition temperatures Tg of about 30-40 oC are found when the same epoxy thermoset is cured at 10 oC, though some post-curing may occur when temperatures are increased again after initial cure. The same principles of Tg development also apply to epoxy thermoset systems without plasticisers. It is therefore motivating to better understand the impact of temperature on mechanical integrity comparing conventional and new curing agents with minimum to no plasticizer.
Figure 7 shows the storage modulus of cured clear coatings as a function of temperature by DMA. The first scan is represented by solid lines versus the second scan by solid line with markers. For CA1 and CA2 based coatings, the second scan curves shifted significantly to higher temperature. Therefore, the glass transition temperature Tg of coatings based on CA1 and CA2 demonstrated a large increase between the first and second scan. Both CA1 and CA2 have first scan Tg of about 57 oC which in the second scan increased to 117oC and 108oC respectively, almost double. This behavior is illustrative for conventional epoxy thermoset systems where the presence of non-reactive plasticizers can exit the coating upon exposure to high temperature. On the contrary, MVB1, MVB2, and MVB3 based coatings show no such shortfall. The increase in Tg between first and second scan is much smaller than that of CA1 and CA2, <15 oC for MVB1, MBV2, and MBV3. Figure 7 clearly demonstrates the improved temperature resistance property of coatings based on new curing agents. This temperature resistance feature is especially beneficial for flooring exposed to high temperatures, such as factories or kitchens, therefore prolongs the service life of a floor and provides cost-saving benefit to floor owner.
New Curing Agents Provide Excellent Adhesion to Concrete and Barrier Property to Moisture Vapor Transmission
In previous sections, we demonstrate the advantages of the new curing agents MVB1, MVB2, and MVB3 which allow coating formulators to design formulations with minimum to no plasticizer. These formulations offer tailorable cure speed, fast property development, and excellent mechanical properties. In this section, we will focus on the benefits of using these new curing agents as a moisture vapor barrier to address the moisture transmission issue in concrete.
Adhesion to Concrete
Adhesion to concrete is one of the key requirements for a moisture vapor barrier coating as it is the first line of defense against the moisture vapor transmission from the concrete slab. The moisture vapor barrier coatings are applied to concrete that can contain varying amount of water depending on the age and the condition of the concrete. The vapor barrier is required to adhere to both dry and damp concrete to avoid any failures of subsequent materials placed or coated on top. Dry and damp concrete in this study are defined as concrete cured in a controlled environment (50% RH/23 oC) greater than 28 days after casting, and green concrete as a fresh concrete cured for 6 days. All concrete blocks had a surface profile of CSP 2 or 3 (International Concrete Repair Institute) as would be recommended for surface preparation. For damp concrete testing, the concrete block was submerged in water for 24 hours, and then removed from water for testing. The relative moisture content was determined by a Testo 606-1 material moisture meter that was set to material number 3 (cement screed, concrete). Dry and damp concrete contains 1.5% and 3% of moisture respectively. The coatings were applied by roller at 10 mil wet film thickness and cured at 50% RH/23 oC for 7 days. Adhesion testing was conducted in accordance with ASTM D7234 method. Figure 7 displays the excellent adhesion of all moisture vapor barrier coatings to dry and damp 28 day cure concrete, and 6 day pour green concrete. Green concrete is an aggressive test as it contains a significant amount of moisture in the concrete and the properties of the concrete are not yet fully developed. Excellent adhesion of moisture vapor barrier coatings to concrete substrates provides a good foundation for the subsequent layer to be coated on.
Carbamation Resistance, Intercoat Adhesion, and Overcoatability
Intercoat adhesion is crucial for a moisture vapor barrier coating since it is a primer and serves as a tie coating between the concrete substrate and the subsequent coatings applied on top. One fundamental study to investigate whether a coating has good intercoat adhesion is a carbamation resistance test. Carbamation occurs when low molecular weight amines from a curing agent migrate from the bulk of the coating to the surface, where they react with carbon dioxide and moisture from the air to form ammonium salts. This process is highly undesirable as it competes with the needed amine-epoxy reaction. It is favored especially at lower temperature and high humidity and manifests itself as haziness, reduced gloss, or white salt on the surface. Carbamation not only reduces the visual appeal of the coating, but also leads to poor intercoat adhesion. Carbamation resistance is critical for a primer to ensure good intercoat adhesion to the subsequent layer coated on top.
Carbamation resistance testing was evaluated using the wet patch method described in ISO 2812. Coatings were applied onto a black Laneta card and exposed to a cotton patch saturated with water for 24 hours following the specified cure time and temperature. Assessment of carbamation follows a relative scale 1-5, as 5 being best with no signs of carbamation. New curing agents were benchmarked against a commercially available fast curing agent used as moisture barrier, Ref1. Table 5 summarizes the carbamation test results at ambient and 10 oC. All new curing agents show good carbmation resistance at ambient temperature. Reference fast curing agent showed very poor result at low temperature of 10 oC.
Overcoatability is another important criterium for a moisture vapor barrier coating (MVB). Generally, most commercial floorings have a multi-layer system to get the highest performance. For a seamless flooring system, a primer coating is followed by a mid-coat and/or topcoat. The mid-coat and topcoat could be filled, pigmented or clear coatings and could be any of the vast thermoplastic or thermoset technologies including epoxies, polyurethanes, polyureas, and acrylics.
For a resilient flooring system, a primer coating is followed by a self-leveling coating such as a cementitious overlay or other thick build coating to smooth out the floor. The final step would be to apply a floor covering such as wood, tile, or carpet.
The overcoatability is determined by the intercoat adhesion of the moisture vapor barrier layer to the subsequent layer following a specified schedule. It also provides information on overcoat window.
Two test methods were employed to assess the intercoat adhesion. In the first test, a commercial aliphatic polyurea was coated at 10 mil thickness on the surface of the 10 mil thick MVB1 or MVB2 coatings after a cure time of 4 hours and 24 hours cure at ambient conditions (50% RH/23oC). The intercoat adhesion test was conducted based on the method described in ASTM D3359 after the system was cured for 7 days cure at 50% RH/23 oC, and the results were reported on a scale of 1-5 with 5 being the best.
The data in Table 6 shows that all MVB coatings provide excellent intercoat adhesion to an aliphatic polyurea topcoat. For fast curing agents MVB2 and MVB3, the topcoat can be applied in 4 hours with excellent intercoat adhesion. This provides a solution for fast return to service where multiple layers can be applied within one day.
Excellent overcoatability is also demonstrated in a thick build cementitious overlay. In this test, a commercial cementitious overlay was applied on top of MVB1 and MVB2 at 1/8-inch thickness using a small trowel after a minimum time of 6 hours and 24 hours cure at 50% RH/23°C. The intercoat adhesion testing was conducted using ASTM D7234 after 14 days cure. The cementitious overlay also showed good intercoat adhesion to the new MVB layer (Table 6).
The overlay has a strong bond to the MVB coatings as the dollie pull leads to cohesive failure of the cementitious overlay instead of the adhesive failure at the interface. For both MVB1 and MVB2, the test was done within 24 hours. It is possible for the MVB coatings to have an overcoat window beyond 24 hours but this was not tested in this study. We also expect MVB3 would perform similarly to MVB2 based on the design principle of the curing agent.
Barrier to Moisture Vapor Transmission
Concrete is a permeable material that allows moisture underneath the concrete, or trapped in the concrete itself, to migrate as a vapor phase to the concrete surface. This moisture migration can cause floor failure and result in significant financial loss. Applying an effective moisture vapor barrier (MVB) on the surface of the concrete can significantly reduce moisture vapor from migrating out of the concrete slab due to hydrostatic pressure or capillary flow. The MVB should have a very low moisture vapor transmission or flow through the coating because even a small amount of moisture collected under the non-permeable floor covering can lead to adhesive failures, delamination, or microbial growth.
One of the most common techniques to measure moisture vapor transmission (MVT) of a coating is using ASTM E96 method. In this method, test specimens at each coating thickness are fabricated to the dimensions of a flanged stainless steel pans as specified in the method. The pans are filled partly with water, then the tested materials are tightly fitted to seal the pans and monitored at controlled environment (23°C/50% RH).
The test specimens are weighed regularly to the nearest 0.0001 gram and this process continues until enough data points are collected to establish a moisture vapor transmission rate through the coating. The permeance value is calculated from the moisture vapor transmission rate in perms (grain of water per hour per square feet per inch of mercury) and the results is recorded for each test specimen. ASTM F3010 certification requires a very low permeance value of 0.1 perm or lower to be an effective moisture
vapor barrier.
The moisture vapor transmission rate of the new curing agents, measured using ASTM E96 method, and benchmarked alongside a 50/50 blend of MVB1 and a benzyl alcohol containing curing agent CA39 and a commercially available fast cure MVB system Ref1. The permeance results are shown in Figure 8. All new MVB curing agents outperform Ref1 with lower permeance at the same thickness of 16 mil or lower thickness of 14 mil. MVB1, MVB2, and MVB3 show two times lower permeance than the MVB1/CA3 blend at the same coating thickness of 16 mils.
Even at very thin thickness of 10 mil, MVB2 and MVB3 display low water vapor transmission to meet the ASTM F3010 certification requirement of 0.1 perms. MVB1/CA3 blend contains about 20% benzyl alcohol and it is hypothesized that some benzyl alcohol could be emitting out of the coating over time leaving voids in the film. The voids could provide channels for moisture vapor to pass through.
Even at low thickness of 10 mils, all new MVB curing agents outperforms MVB1/CA3 at 16 mil. The moisture vapor testing indicates that both MVB1, MVB2, and MVB3 can be utilized as an effective moisture vapor barrier due to very low permeance to water vapor. And the high performance at lower thickness translates to cost-saving for end-users.
Conclusions
We have demonstrated in this paper epoxy systems using new amine curing agent technology that offers tailorable cure speed, fast property development and good carbamation resistance. The new technology enables the design of epoxy systems with minimum to no plasticizer. Conventional cycloaliphatic amine-cured epoxies without plasticizers can suffer from incomplete conversion and poor performance. The new amine curing agents presented in this paper, MVB1, MVB2 and MVB3, containing minimum to no plasticizer, deliver high conversion and better mechanical property. They provide an alternative to conventional plasticizer containing curing agents to meet compliance with stringent emission requirement.
The paper details fundamental study of new curing agents MVB1, MVB2 and MVB3, against conventional plasticizer containing curing agents CA1 and CA2 in flooring applications. Various analytical techniques were employed to understand the cure behavior of these curing agents. DSC, DMA and near infrared data corroborated with each other to confirm high reaction conversion of these new epoxy systems, comparable to conventional plasticizer containing system using CA1 and CA2. Minimizing or eliminating plasticizer in MVB1, MVB2 and MVB3 did not result in negative effects of low conversion and compromised mechanical properties. On the contrary, compressive testing showed MVB1, MVB2 and MVB3 provide coatings with higher compressive strength and toughness compared to CA1 and CA2. In addition, DMA test demonstrated minimizing plasticizer imparted better high temperature resistance.
Furthermore, MVB1, MVB2 and MVB3 provide outstanding adhesion to dry, damp and green concrete. All showed excellent intercoat adhesion to organic coatings such as polyurea, or thick cementitious overlayer. Mostly importantly, MVB1, MVB2 and MVB3 are proven to be excellent barriers to moisture vapor transmission at coating thickness of 10 mil and have been used as excellent moisture vapor barriers to address moisture vapor transmission in concrete.
The performance features demonstrated in this paper show that MVB1, MVB2 and MVB3 are great products for epoxy floor coatings to meet stringent emission requirement, more importantly they are excellent choice for moisture vapor barrier to meet ASTM F3010 certification of less than 0.1 perm even at 10 mil thickness.
Acknowledgements
The authors would like to thank Mike Oberlander, Dave Brown, and Edwin Lijffijt for their contributions in the experimental work presented in this paper.
References
1. “Moisture in Concrete and Moisture-sensitive Finishes and Coatings” in Cement Concrete & Aggregates Australia April 2007.
2. Tanaka, Y., “Synthesis and characteristics of epoxides”, in C.A. May, ed., “Epoxy Resins Chemistry and Technology” Marcel Dekker, 1988
3. Wissanrakkit, G. and Gilham, J.K., J. of Coatings Technology, 1990, 62 (783), 35-36.
4. Abbreviations of amine curing agents and further details are included in the respective technical datasheets from Evonik Corporation: Ancamine® 2739 curing agent (MVB1), Ancamine® 2800 curing agent (MVB2), Ancamine® 2850 curing agent (MVB3), Ancamine® 1618 curing agent (CA1), and Ancamine® 2519 curing agent (CA2).
5. Epodil® 748 Reactive Diluent, Evonik Corporation.
6. (a) Lee, W. I., Loos, A. C., Springer, G. S. J. Composite Mat. 1982, 16, 510. (b) Dangayach, K., Smith, C. J. Coatings Technology, 1996, 68 (863), 29-37.
7. Vratsanos, M. Coatings Tech July 2017, 28-38.
8. (a) Xu, L.; Fu, J. H.; Schlup, J. R. J. Am. Chem. Soc. 1994, 116, 2821-2826; (b) Fu, J. H.; Schlup, J. R. J. Appl. Polymer Sci. 1993, 49, 219-227.
9. Ancamine® 2764 curing agent (CA3).