Ingrid K. Meier, K. Michael Peck, and Christine Louis , Evonik Corporation03.13.20
Abstract
Claude Monet once commented that “Color is my day-long obsession, joy and torment” – a sentiment that resonates with formulators of waterborne architectural paints who need to design base paints that can accept a variety of liquid colorants, enabling a paint line with the broadest possible color palette.
With the advent of point-of-sale (POS) tinting systems, many paint manufacturers now produce only a small number of base paints to which the colorants are added at the store. This allows paint manufacturers and stores to manage inventories by stocking fewer materials, and it significantly decreases the amount of unused paint that goes to waste each year.
However, the colorants used in POS tinting systems must have excellent compatibility with different base paints to ensure consistent, reproducible colors under all conditions. This remains one of the toughest challenges facing the waterborne architectural paint formulator today.
Difficulties occur when additives used to stabilize the pigments in the colorant interact with additives in the base paint, causing loss of pigment stabilization and flocculation. This leads to an immediate or gradual color change that can be observed in a “rub-out” test in the laboratory. “Problem pigments” (like PV23, PBk7 and PB15:3) give rise to the most challenges with regard to colorant acceptance. This paper describes the mechanisms involved in colorant stabilization and, using chemical structure-property relationships, explains how specific surface active agents can be used within the base paint formulation to prevent destabilization of the colorant, ensuring that it performs as intended.
Introduction
Point-of-Sale Tinting Systems: Benefits and Challenges
Point-of sale (POS) tinting systems allow paint manufacturers to produce only a few base paints into which colorants are added at the store. This approach enables customers to decorate with a very broad color palette while paint manufacturers and retailers can save money by stocking fewer materials. As a result, much less unused paint goes to waste. However, the colorants used in POS tinting systems must have excellent compatibility with different base paints to ensure consistent, reproducible colors under all conditions.1, 2
Retailers who sell architectural paints typically use computer-controlled automated dispensing machines that hold up to 16 different colorants and simultaneously dispense up to four POS colorants. Larger scale machines, while less frequently used, are even more flexible and efficient. Most dispensing machine software can now interface with a spectrophotometer as well as large databases, enabling many retailers to “read” a paint chip and calculate the exact recipe of colorants required to be dispensed into the selected base paint in order to achieve a near-perfect color match. Oftentimes universal colorants are used in these POS tinting machines because they can be used to tint both waterborne and older solvent-containing and oil-based paint formulations.3, 4
Increasingly stringent regulations of the VOC content of paints and colorants have driven reformulation of universal colorants so that they, too, are now very low or “near zero” VOC. While these new universal colorants can be used to achieve nearly identical colors to those achieved using the higher-VOC technology, the lower-VOC colorants use different additives to achieve maximum color strength and dispersion stability. Thus, these low-VOC colorants may perform differently in both dispensing machines and the base paints.
Some large paint manufacturers control the POS tinting systems and machines used to color their paints, and these companies have developed their own colorant technologies in order to ensure that “near-zero” VOC colorants are added to their low- or zero-VOC paints. Other paint manufacturers must rely on the retailer to use universal colorants produced by other suppliers in order to tint their base paints. Changes in the universal colorant compositions will often necessitate a reformulation of the base paints in order to compensate for any adverse effects the new colorants may have on final paint properties including color development and colorant acceptance.
Color Development and Colorant Acceptance
The term “color development” describes the degree of tint strength and color quality achieved when a paint is tinted. Good color development implies that the color has not changed and is of the expected color strength. “Colorant acceptance” is the ability of a base paint to be tinted in a manner that enables consistently good color development to be achieved uniformly throughout the applied coating. A practical method for determining the degree of colorant acceptance is the so-called “rub-out” test in which an area in the drying (tacky) paint film is rubbed and, once dry, the color properties of the rubbed and unrubbed areas are measured and compared. Any color differences can be readily detected and are indicative of destabilization of at least one of the pigments used in the formulation.
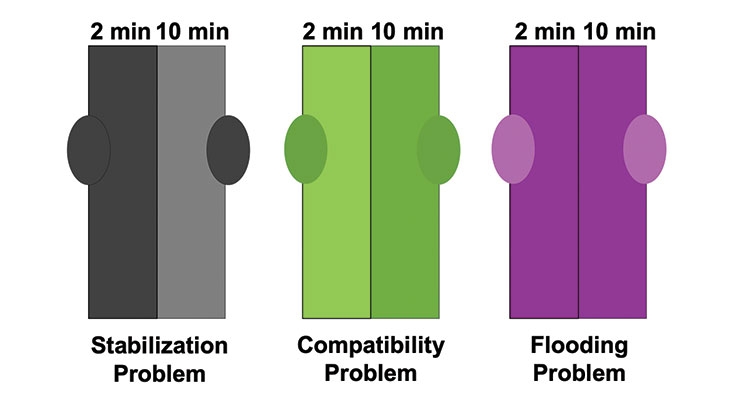
Difficulties occur when additives used to stabilize the pigments in the colorant interact with the pigments and fillers in the base paint. This migration of additives to and from pigments and fillers can result in under-stabilized particle surfaces. This phenomenon can cause loss of pigment stabilization and flocculation of pigment particles which can lead to an immediate or gradual color change. “Problem pigments” like carbazole dioxazine violet, carbon black, and phthalocyanine blue and green are often mentioned by paint formulators as posing the most challenges with regards to colorant acceptance.5 Figure 1 illustrates some typical colorant acceptance issues that are seen in “rub-outs” of tinted paints using colorants that become destabilized on addition to a base paint. Note that each paint has been drawn down after two minutes (left side) as well as after ten minutes (right side) of shaking the tinted paint on a Skandex shaker. The two drawdowns on the far left in Figure 1 show a stabilization problem that results because the TiO2 and/or filler(s) in the base paint is under-stabilized. After the colorant is added and shaken, the surfactant/dispersant package that had been stabilizing the pigment in the colorant is “stolen” to better stabilize the white. The result is under-stabilization of the pigment from the colorant, leading to flocculation and lower color strength. The two drawdowns in the example in the middle of Figure 1 show a compatibility problem. The base paint and colorant are compositionally very different from each other and are likely to differ substantially in polarity. Longer shaking times are required to achieve effective mixing and full color development, as is seen in the increased color strength with increased mixing time. Finally, the example on the far right of Figure 1 illustrates a pigment flooding problem. The differences in pigment mobility within the paint give rise to separation of the pigments. Typically, organic pigments rise to the surface due to their lower density. This phenomenon is not impacted by shaking time, and the true color is revealed by rub out.
Surfactants: The Cause of and Potential Solution to the Problem
The term “surface active agents” is a broad one that describes a wide variety of amphiphilic molecules; these molecules are often referred to as “surfactants” – a contraction derived from “surface active agents.” A commonality amongst all surfactants is that at least one hydrophilic and at least one hydrophobic functional group exists within a single lower-molecular-weight molecule or higher-molecular-weight polymer. In aqueous formulations, these amphiphilic molecules are driven out of the water in order to minimize the energy of the system. This behavior results in many surfactants forming micelles when their concentration gets high enough that the hydrophobic groups can pack within the interior of the aggregate so that only the hydrophilic groups are exposed to the water. In a coating formulation, the hydrophobic group(s) on a surfactant may also adsorb onto the surfaces of pigment and filler particles. Further molecular design can be used to tailor the hydrophobes within a surfactant to be more pigment-affinic and the hydrophiles to provide steric and/or electrostatic stabilization, enabling the surfactant to act as a dispersant.
Surfactants are, therefore, the additives of choice for providing the wetting, dispersion and stabilization of the pigments and fillers used in waterborne coatings.6 Lower-molecular-weight surfactants with only one or two shorter hydrophobic groups are typically used to provide dry particle wetting and to enable the efficient milling needed to achieve optimal pigment particle sizes. Higher-molecular-weight, oligomeric or polymeric surfactants are most often used to provide longer term stabilization due to their having multiple pigment-affinic and electrostatic or steric stabilizing groups within a single molecule.7
Nonionic surfactants are widely used in coatings, and it is now understood that structural differences in their molecular architectures cause them to behave very differently. For example, low-molecular-weight, highly-branched, relatively hydrophobic surfactants function well as dynamic wetting agents; whereas higher-molecular-weight, highly-ethoxylated alcohol alkoxylates form micelles in aqueous systems and function as emulsifiers and co-dispersants.7 In order to better understand the behavior of surfactants in coating formulations, a series of experiments was conducted to study the ability of various nonionic surfactants to improve colorant acceptance in waterborne architectural coatings.
Results and Discussion
Experimental
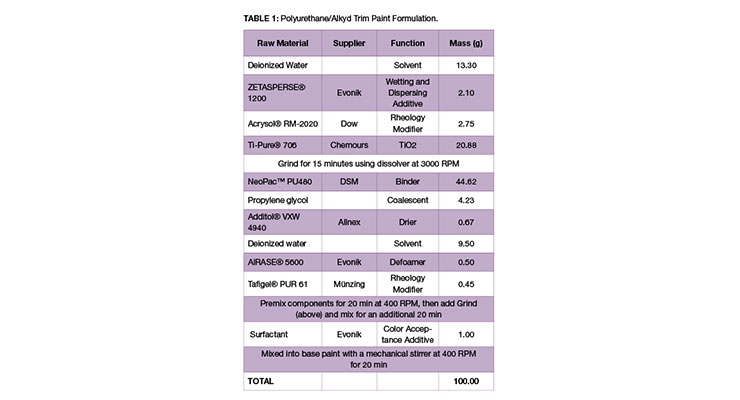
Commercial waterborne base paints were purchased from local retailers and used as received; these included an acrylic latex extra opaque wall and ceiling matte white paint from Praxis (Sencys Muur & Plafond Extra Dekkend Mat Wit) and a styrene-acrylic base paint from Avis Professional (Flevotine Interieurlatex). Additionally, a polyurethane/alkyd trim paint was prepared using the formulation shown in Table 1. The following colorants were obtained from Elementis and Chromaflo Technologies: TINT-AYD® AQ 10 HC (PR112), TINT-AYD WCH Plus 3 (PBk7), Colortrend 807-7055 EXE ES Blue (PB15:3) and Colortrend 807-8894 JXE HS (PV23); all were used as received.
The acetylenic diol alkoxylates (TMDD-1EO, TMDD-4EO, TMDD-10EO, TMDD-23EO and TMDD-EOPO), Anionic/Nonionic Formulated and Formulated Nonionic surfactants, and alcohol ethoxylates (AE HLB 7.5, AE HLB 10.6, AE HLB 10.7, AE HLB 13.1, AE HLB 13.9, AE HLB 14.4, AE HLB 18.2 and Highly Branched AE HLB 17.9) that were evaluated as Color Acceptance Additives were commercial products sold by Evonik Corporation, and they were used as received. Each was post-added to the base paint at 1.0 wt% (as supplied on total formulation) and mixed into the base paint using a mechanical stirrer for 20 minutes at 400 RPM. The paint was allowed to equilibrate overnight and then 1.0 wt% of colorant was added to the base paint and the paint was shaken using a Skandex shaker SO-20a for two minutes. After two minutes a small sample was removed for evaluation. The remaining paint was then mixed for an additional eight minutes on the Skandex shaker for a total mixing time of ten minutes.
Drawdowns of both the “2 min” and “10 min” paint samples were made on a plain white Leneta chart using an automated drawdown machine with bar designed to apply a 150-micron wet film thickness; rub-out tests were performed one minute after drawdown using a finger to make a circular rub-out in the drying paint.
To determine the influence of the additives on color compatibility and color strength, L*, a*, b*, c*, h* and ∆E* values were measured on the dried drawdowns using an X-Rite 939 Spectrodensitometer using a D65/10° illuminant/observer combination and an 8 mm measuring aperture. The tinted paint containing no post-added surfactant was used as the reference, and its color strength was set to 100%. Each drawdown was evaluated to determine: color strength after two and ten minutes of shaking, rub-out test after two and ten minutes of shaking, and ∆E between the drawdowns obtained after two and ten minutes of shaking. The ∆E* values less than 0.5 were considered to show no color difference; 0.5 < ∆E* < 1.0 were only slightly visible, while ∆E* values greater than 1.0 were deemed to be unacceptable.
Paint viscosities were measured using a Brookfield DV-I Prime viscometer, spindle 64 at 100 RPM. Sag resistance was determined using an Erichsen Sag Tester Model 419 (ASTM
D 4400 version).
Results
Initial screening studies were first conducted in order to determine whether any trends were immediately obvious as well as to get better ideas for future experimental direction. In these screenings, a broad number of surfactants and dispersants were post-added at 1.0 wt% to a base paint; once thoroughly mixed, the base paint was tinted using a designated dose of colorant. Because of the breadth of chemistries evaluated, top performers for a given base paint-colorant system could be identified; however, a systematic structure-property understanding was not possible.
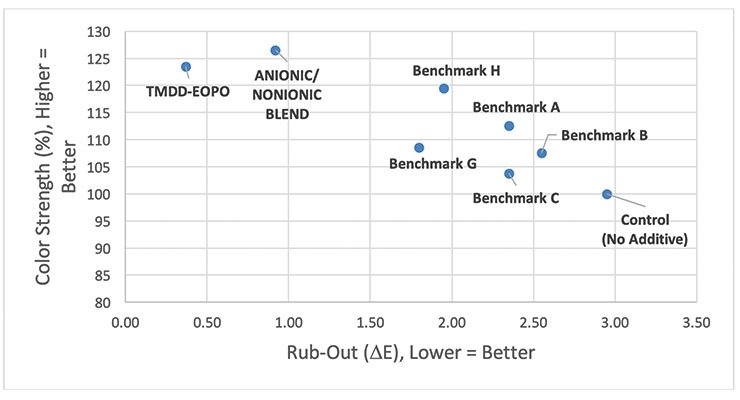
For example, in a waterborne acrylic base paint that was tinted using 1.0 wt% of a pigment violet 23 colorant, it was observed that many surfactants of varying chemistries were able to slightly to significantly improve the color strength of the paint; however, only the alkoxylated acetylenic diol (TMDD-EOPO) showed a significant improvement (∆E < 0.5) in the rub-out test (Figure 2).
While there is a very slight improvement in both color strength and rub-out (∆E) when Benchmark C (a potassium alkyl phosphate ester) is added, the significant improvements observed when the TMDD-EOPO additive is used is noteworthy (Figure 3).
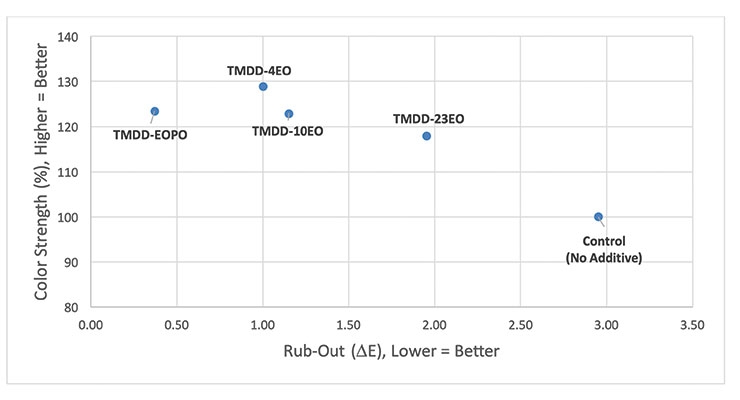
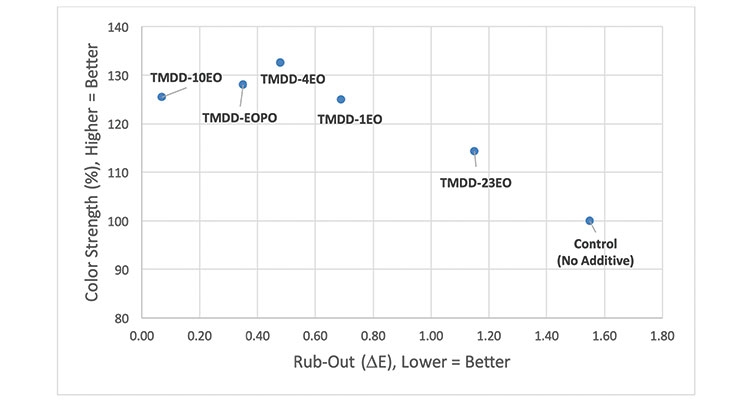
Because of the positive improvements in color acceptance that were seen for the TMDD-EOPO, additional screening tests were conducted to determine what effects type and degree of alkoxylation have on color strength and rub-out. To do this, a series of acetylenic diol ethoxylates (TMDD-4EO, TMDD-10EO and TMDD-23EO) were evaluated as 1.0 wt% post-additions to the same waterborne acrylic paint used in the previous study, and the color strength and rub-out data after tinting with 1.0 wt% pigment violet 23 colorant was compared to that obtained for the control (no additive) and TMDD-EOPO (Figure 4).
All the alkoxylated acetylenic diols significantly improve the color strength obtained on tinting the acrylic base paint with 1.0 wt% pigment violet 23 colorant. This is not surprising because it is well known that dynamic wetting agents can enable more efficient wetting of pigment particles in order to enable better dispersion of the pigment particles; as long as the polymeric dispersant present in the formulation can stabilize these smaller pigment particles, higher color strength results.6 As the degree of acetylenic diol ethoxylation decreases, the color strength increases. This correlates with previous observations that acetylenic diol ethoxylates with higher degrees of ethoxylation are less efficient at lowering dynamic surface tension than their low-mole ethoxylate analogues. The tendency of these highly branched wetting agents to resist being adsorbed on particle surfaces ensures that more of the molecules are available to provide this needed dynamic wetting benefit.8-10 Interestingly, within the series of three acetylenic diol ethoxylates, both color strength and rub-out (∆E) values improve to more significant extents as degree of ethoxylation decreases. That said, only TMDD-EOPO, which has a degree of ethoxylation in between that of TMDD-4EO and TMDD-10EO but also has been hydrophobically modified via propoxylation, is able to render this base paint robust enough to tinting with the PV23 colorant (i.e., rub-out ∆E < 0.5).
A similar study was conducted using the same waterborne acrylic base paint but tinting it with 1.0 wt% of a pigment red 112 colorant. The results are shown in Figure 5. Here, again, the simple presence of 1.0 wt% of the dynamic wetting agents is enough to provide a 15% to > 20% increase in color strength. However, the improvement in rub-out values varies with degree of alkoxylation. Interestingly, the additive with the highest level of ethoxylation (TMDD-23EO) provides only somewhat better colorant acceptance while lower levels of ethoxylation show even better colorant acceptance. Here there appears to be a “sweet spot,” with TMDD-10 (the ~ 10 mole ethoxylate) providing the optimal colorant acceptance.
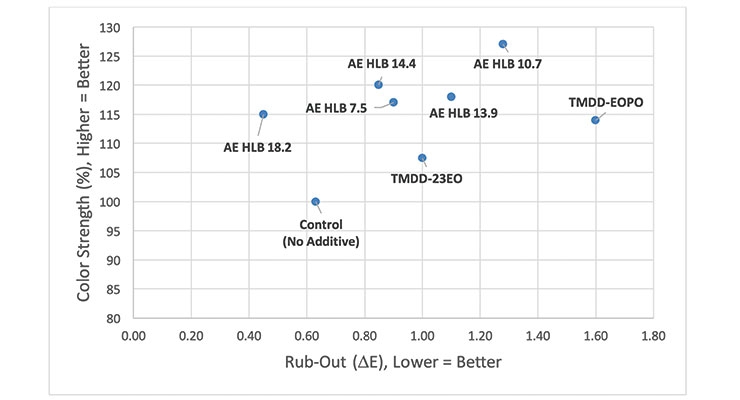
In order to understand whether similar trends would translate to base paints and colorants of different chemistries, a waterborne styrene-acrylic base paint that was tinted using 1.0 wt% of a pigment blue 15:3 colorant was studied. Tinting this formulation with the PB15:3 colorant resulted in a slight rub-out difference (∆E ~ 0.6); however, the paint’s color strength could be improved significantly with the use of a wide variety of surfactants. That said, many of the additives tested, including the alkoxylated acetylenic diols, adversely impacted the colorant acceptance, resulting in ∆E values > 1 (Figure 6). In this system, a series of linear alcohol ethoxylates of varying hydrophile-lipophile balance (HLB) values was also studied.
While these alcohol ethoxylates represent a broad distribution of molecules exhibiting several different hydrophobic groups (i.e., of different carbon number) as well as varying degrees of ethoxylation, HLB values can be a useful way to express a surfactant’s overall physico-chemical characteristics. A surfactant with a higher HLB value is more hydrophilic in nature than those with lower HLB values.11
As shown in Figure 6, the base paint has good color acceptance already and only the highest HLB alcohol ethoxylate (AE HLB 18.2) can improve both the color strength and the rub-out (∆E) for this base paint-colorant system. This is likely due to the ability of AE HLB 18.2 to provide steric stabilization – acting as a co-dispersant – to improve the overall stabilization of the white pigments and fillers in the styrene-acrylate base paint. In such a formulation, the styrene-acrylic binder also functions as a polymeric dispersant for the particles, operating via an electrostatic stabilization mechanism.
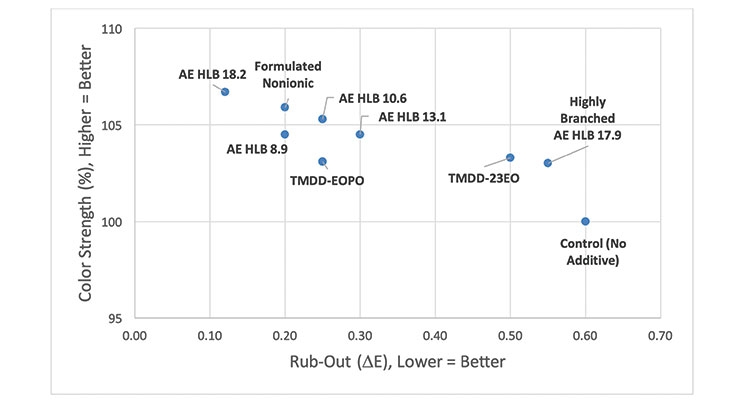
Because the behavior of surfactants in base paints varies greatly depending on the chemical natures of the binder as well as the other components of the base paint, it was decided to conduct an evaluation of surfactants in a polyurethane/alkyd trim paint formulation in which most of the chemical composition could be understood. Therefore, the base paint formulation shown in Table 1 (Experimental) was prepared. In this formulation, the dry titanium dioxide was ground with a formulated dispersant that contains an anionic polymeric dispersant (providing electrostatic stabilization) as well as two nonionic surfactants (providing dynamic wetting and temporary steric stabilization). 1.0 wt% of each Color Acceptance Additive was post-added as in previous studies, and, finally, each base paint was tinted using 5.0 wt% carbon black colorant (PBk7). A comparison of color strength and rub-out (∆E) values is shown in Figure 7.
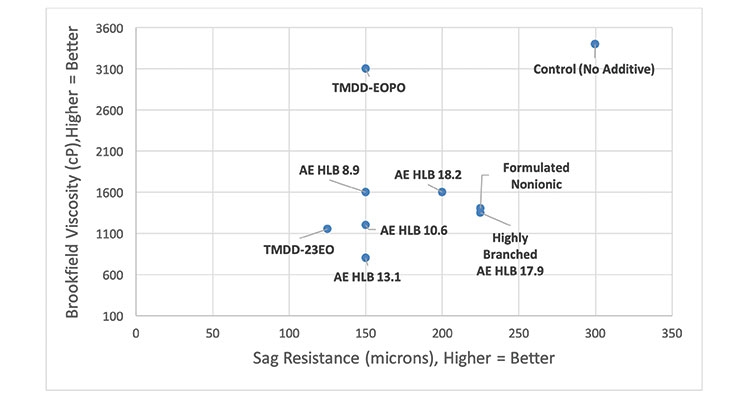
This base paint appears to be adequately stabilized already, but the addition of 1.0 wt% alcohol ethoxylate or alkoxylated acetylenic surfactant can still improve the color strength by 3-7% relative to the control (no additive). Interestingly, significant improvement in rub-out (∆E) values does not appear to strictly correlate with the HLB value for the alcohol ethoxylates. Moreover, many of these surfactants affect viscosity and sag resistance upon tinting, as can be seen in Figure 8. Therefore, an additive that improves color strength, rub-out and has less of an impact on viscosity and sag resistance would be the best option. In this regard, the Formulated Nonionic Surfactant and the AE HLB 18.2 appear to be the better choices amongst the surfactants studied here (Figure 9). Clearly the complex nature of coating formulations and the variety of interactions that are available to surface active molecules makes it rather difficult to develop a robust predictive model that can be used to identify the best additive for improving color acceptance without adversely affecting other properties.
Discussion
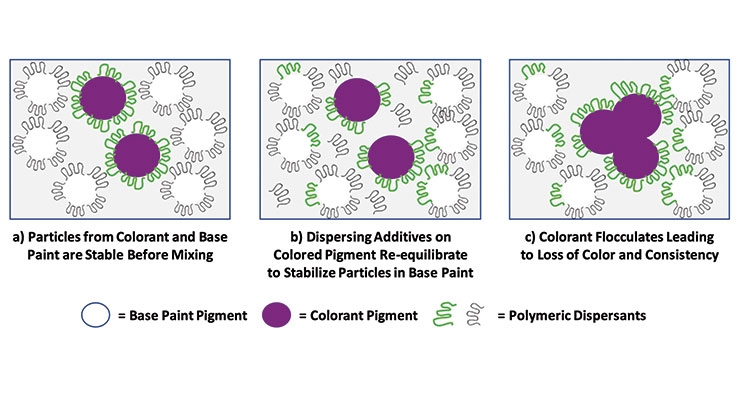
Color development in a tinted base paint is dependent upon the stability of the pigments and fillers dispersed in both the base paint and the colorants. When the colorants and paint are mixed, the additives present in each component can re-equilibrate between all pigments and fillers present in the formulation. Many base paints, such as the waterborne acrylic base paint studied here, use the minimum quantity of additives needed to stabilize the titanium dioxide and fillers in the base paint. Tinting these under-stabilized base paints often results in a re-distribution of additives – removing them from the colored pigments that come from the colorant in order to satisfy the additive-starved particles present in the base paint. Colorant destabilization and loss of color strength result (Figure 10).
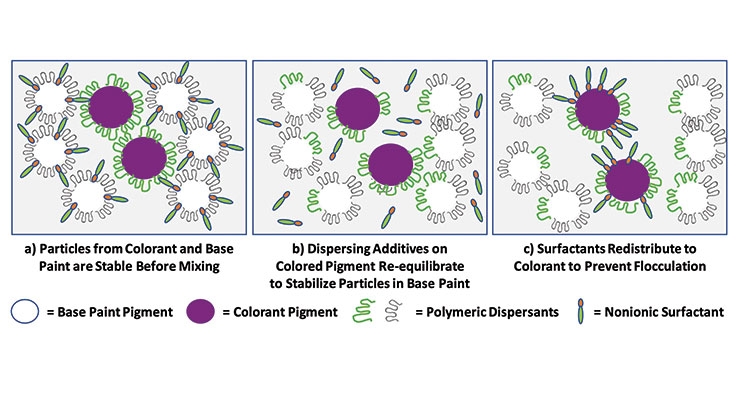
The formulation of pigment dispersions used in both base paints and colorants can be enhanced by using the optimal surface active agents to wet, disperse and stabilize the solid particles in the medium as well as to provide improved performance of the paint during application. As can be seen from the composition of the dispersant used in the waterborne polyurethane/alkyd base paint in Table 1, most dispersions employ at least two, and often three or more, surface active components that are combined in order to achieve the desired properties. These surfactants can also help to improve the compatibility of the dispersion when it is letdown into another system. Similarly, they can help minimize shock and color acceptance issues when a colorant is added to a base paint during tinting. It is believed that these surfactants function by migrating to the unprotected pigment surfaces created when the dispersing additives that are originally protecting the pigment re-distribute on mixing. Thus, these surfactants can prevent the destabilization of the pigments and lead to excellent colorant acceptance (Figure 11).
Conclusions
The experiments conducted in this study have demonstrated that nonionic surfactants, like alkoxylated acetylenic diols, that lower dynamic surface tension are also often effective at increasing the color strength achievable on tinting a given base paint. Additionally, alcohol ethoxylates – particularly those with higher HLB values – are often able to improve color acceptance presumably because of their ability to provide steric stabilization to pigments and fillers. In this way, the high-mole alcohol ethoxylates can function as co-dispersants when used in combination with acrylic or styrene-acrylic binders that provide the needed anionic stabilization. All these additives can stabilize the pigments during surfactant/dispersant re-equilibration after mixing. However, some formulations see improvements in color acceptance when higher-mole alkoxylates are used while other paints require the more hydrophobic lower-mole alkoxylates to see such improvements. It is likely that the chemistries employed in both the base paint and the colorants play significant roles in determining what is needed to stabilize all particles in the combined formulation; therefore, predictability
remains elusive.
References
1. S. Heutz, M.N.S. Kunnen and B.A. Wilbanks, Customized Tinting System Design: Advanced Tinting Systems – An Integrated Concept, Proceedings of the 39th Annual Waterborne High-Solids, and Powder Coating Symposium, 2012, DEStech Publications, Inc., Lancaster, PA, 617-627.
2. M. Kunnan and S. Heutz, Integrated Tinting Systems, Paint and Coatings Industry, 2011 (http://www.pcimag.com/articles/93432-integrated-tinting-systems).
3. Fluid Management Inc., Wheeling, IL, USA (http://www.fluidman.com).
4. COROB S.p.A., San Felice Sul Panaro MO, Italy (http://www.corob.com).
5. C. Louis, Improving Colourant Compatibility and Colour Development in Architectural Coatings, Pitture e Vernici - European Coatings – Formulation, 2016, 5, 22-28.
6. K.M. Peck, Formulating Optimized Aqueous Dispersions Using Surface Active Additive Triangulation, Journal of Coatings Technology, 2014, 50-57.
7. K.M. Peck, The Role of Surfactants in Aqueous Pigment Dispersion, Paint and Coatings Industry, 2016, 62-66.
8. J. Schwartz, The Importance of Low Dynamic Surface Tension in Waterborne Coatings, Journal of Coatings Technology, 1992, 64, 65-74.
9. S.W. Musselman and S. Chander, Adsorption of Acetylenic Diol-Based Nonionic Surfactants on Lampblack and Phthalocyanine Blue Pigment, Journal of Colloid and Interface Science, 2002, 256, 91-99.
10. J.A. Marsella, C.J. Mammarella and I.K. Meier, Interactions of surfactants with a derivatized low molecular weight styrene maleic anhydride co-polymer – differences between acetylenic diol-based wetting agents and other ethoxylates, Colloids and Surfaces A: Physicochem. Eng. Aspects, 2004, 234, 63-69.
11. W.C. Griffin, Classification of Surface-Active Agents by “HLB”, Journal of the Society of Cosmetic Chemists, 1949, 5, 311-326.
Claude Monet once commented that “Color is my day-long obsession, joy and torment” – a sentiment that resonates with formulators of waterborne architectural paints who need to design base paints that can accept a variety of liquid colorants, enabling a paint line with the broadest possible color palette.
With the advent of point-of-sale (POS) tinting systems, many paint manufacturers now produce only a small number of base paints to which the colorants are added at the store. This allows paint manufacturers and stores to manage inventories by stocking fewer materials, and it significantly decreases the amount of unused paint that goes to waste each year.
However, the colorants used in POS tinting systems must have excellent compatibility with different base paints to ensure consistent, reproducible colors under all conditions. This remains one of the toughest challenges facing the waterborne architectural paint formulator today.
Difficulties occur when additives used to stabilize the pigments in the colorant interact with additives in the base paint, causing loss of pigment stabilization and flocculation. This leads to an immediate or gradual color change that can be observed in a “rub-out” test in the laboratory. “Problem pigments” (like PV23, PBk7 and PB15:3) give rise to the most challenges with regard to colorant acceptance. This paper describes the mechanisms involved in colorant stabilization and, using chemical structure-property relationships, explains how specific surface active agents can be used within the base paint formulation to prevent destabilization of the colorant, ensuring that it performs as intended.
Introduction
Point-of-Sale Tinting Systems: Benefits and Challenges
Point-of sale (POS) tinting systems allow paint manufacturers to produce only a few base paints into which colorants are added at the store. This approach enables customers to decorate with a very broad color palette while paint manufacturers and retailers can save money by stocking fewer materials. As a result, much less unused paint goes to waste. However, the colorants used in POS tinting systems must have excellent compatibility with different base paints to ensure consistent, reproducible colors under all conditions.1, 2
Retailers who sell architectural paints typically use computer-controlled automated dispensing machines that hold up to 16 different colorants and simultaneously dispense up to four POS colorants. Larger scale machines, while less frequently used, are even more flexible and efficient. Most dispensing machine software can now interface with a spectrophotometer as well as large databases, enabling many retailers to “read” a paint chip and calculate the exact recipe of colorants required to be dispensed into the selected base paint in order to achieve a near-perfect color match. Oftentimes universal colorants are used in these POS tinting machines because they can be used to tint both waterborne and older solvent-containing and oil-based paint formulations.3, 4
Increasingly stringent regulations of the VOC content of paints and colorants have driven reformulation of universal colorants so that they, too, are now very low or “near zero” VOC. While these new universal colorants can be used to achieve nearly identical colors to those achieved using the higher-VOC technology, the lower-VOC colorants use different additives to achieve maximum color strength and dispersion stability. Thus, these low-VOC colorants may perform differently in both dispensing machines and the base paints.
Some large paint manufacturers control the POS tinting systems and machines used to color their paints, and these companies have developed their own colorant technologies in order to ensure that “near-zero” VOC colorants are added to their low- or zero-VOC paints. Other paint manufacturers must rely on the retailer to use universal colorants produced by other suppliers in order to tint their base paints. Changes in the universal colorant compositions will often necessitate a reformulation of the base paints in order to compensate for any adverse effects the new colorants may have on final paint properties including color development and colorant acceptance.
Color Development and Colorant Acceptance
The term “color development” describes the degree of tint strength and color quality achieved when a paint is tinted. Good color development implies that the color has not changed and is of the expected color strength. “Colorant acceptance” is the ability of a base paint to be tinted in a manner that enables consistently good color development to be achieved uniformly throughout the applied coating. A practical method for determining the degree of colorant acceptance is the so-called “rub-out” test in which an area in the drying (tacky) paint film is rubbed and, once dry, the color properties of the rubbed and unrubbed areas are measured and compared. Any color differences can be readily detected and are indicative of destabilization of at least one of the pigments used in the formulation.
Difficulties occur when additives used to stabilize the pigments in the colorant interact with the pigments and fillers in the base paint. This migration of additives to and from pigments and fillers can result in under-stabilized particle surfaces. This phenomenon can cause loss of pigment stabilization and flocculation of pigment particles which can lead to an immediate or gradual color change. “Problem pigments” like carbazole dioxazine violet, carbon black, and phthalocyanine blue and green are often mentioned by paint formulators as posing the most challenges with regards to colorant acceptance.5 Figure 1 illustrates some typical colorant acceptance issues that are seen in “rub-outs” of tinted paints using colorants that become destabilized on addition to a base paint. Note that each paint has been drawn down after two minutes (left side) as well as after ten minutes (right side) of shaking the tinted paint on a Skandex shaker. The two drawdowns on the far left in Figure 1 show a stabilization problem that results because the TiO2 and/or filler(s) in the base paint is under-stabilized. After the colorant is added and shaken, the surfactant/dispersant package that had been stabilizing the pigment in the colorant is “stolen” to better stabilize the white. The result is under-stabilization of the pigment from the colorant, leading to flocculation and lower color strength. The two drawdowns in the example in the middle of Figure 1 show a compatibility problem. The base paint and colorant are compositionally very different from each other and are likely to differ substantially in polarity. Longer shaking times are required to achieve effective mixing and full color development, as is seen in the increased color strength with increased mixing time. Finally, the example on the far right of Figure 1 illustrates a pigment flooding problem. The differences in pigment mobility within the paint give rise to separation of the pigments. Typically, organic pigments rise to the surface due to their lower density. This phenomenon is not impacted by shaking time, and the true color is revealed by rub out.
Surfactants: The Cause of and Potential Solution to the Problem
The term “surface active agents” is a broad one that describes a wide variety of amphiphilic molecules; these molecules are often referred to as “surfactants” – a contraction derived from “surface active agents.” A commonality amongst all surfactants is that at least one hydrophilic and at least one hydrophobic functional group exists within a single lower-molecular-weight molecule or higher-molecular-weight polymer. In aqueous formulations, these amphiphilic molecules are driven out of the water in order to minimize the energy of the system. This behavior results in many surfactants forming micelles when their concentration gets high enough that the hydrophobic groups can pack within the interior of the aggregate so that only the hydrophilic groups are exposed to the water. In a coating formulation, the hydrophobic group(s) on a surfactant may also adsorb onto the surfaces of pigment and filler particles. Further molecular design can be used to tailor the hydrophobes within a surfactant to be more pigment-affinic and the hydrophiles to provide steric and/or electrostatic stabilization, enabling the surfactant to act as a dispersant.
Surfactants are, therefore, the additives of choice for providing the wetting, dispersion and stabilization of the pigments and fillers used in waterborne coatings.6 Lower-molecular-weight surfactants with only one or two shorter hydrophobic groups are typically used to provide dry particle wetting and to enable the efficient milling needed to achieve optimal pigment particle sizes. Higher-molecular-weight, oligomeric or polymeric surfactants are most often used to provide longer term stabilization due to their having multiple pigment-affinic and electrostatic or steric stabilizing groups within a single molecule.7
Nonionic surfactants are widely used in coatings, and it is now understood that structural differences in their molecular architectures cause them to behave very differently. For example, low-molecular-weight, highly-branched, relatively hydrophobic surfactants function well as dynamic wetting agents; whereas higher-molecular-weight, highly-ethoxylated alcohol alkoxylates form micelles in aqueous systems and function as emulsifiers and co-dispersants.7 In order to better understand the behavior of surfactants in coating formulations, a series of experiments was conducted to study the ability of various nonionic surfactants to improve colorant acceptance in waterborne architectural coatings.
Results and Discussion
Experimental
Commercial waterborne base paints were purchased from local retailers and used as received; these included an acrylic latex extra opaque wall and ceiling matte white paint from Praxis (Sencys Muur & Plafond Extra Dekkend Mat Wit) and a styrene-acrylic base paint from Avis Professional (Flevotine Interieurlatex). Additionally, a polyurethane/alkyd trim paint was prepared using the formulation shown in Table 1. The following colorants were obtained from Elementis and Chromaflo Technologies: TINT-AYD® AQ 10 HC (PR112), TINT-AYD WCH Plus 3 (PBk7), Colortrend 807-7055 EXE ES Blue (PB15:3) and Colortrend 807-8894 JXE HS (PV23); all were used as received.
The acetylenic diol alkoxylates (TMDD-1EO, TMDD-4EO, TMDD-10EO, TMDD-23EO and TMDD-EOPO), Anionic/Nonionic Formulated and Formulated Nonionic surfactants, and alcohol ethoxylates (AE HLB 7.5, AE HLB 10.6, AE HLB 10.7, AE HLB 13.1, AE HLB 13.9, AE HLB 14.4, AE HLB 18.2 and Highly Branched AE HLB 17.9) that were evaluated as Color Acceptance Additives were commercial products sold by Evonik Corporation, and they were used as received. Each was post-added to the base paint at 1.0 wt% (as supplied on total formulation) and mixed into the base paint using a mechanical stirrer for 20 minutes at 400 RPM. The paint was allowed to equilibrate overnight and then 1.0 wt% of colorant was added to the base paint and the paint was shaken using a Skandex shaker SO-20a for two minutes. After two minutes a small sample was removed for evaluation. The remaining paint was then mixed for an additional eight minutes on the Skandex shaker for a total mixing time of ten minutes.
Drawdowns of both the “2 min” and “10 min” paint samples were made on a plain white Leneta chart using an automated drawdown machine with bar designed to apply a 150-micron wet film thickness; rub-out tests were performed one minute after drawdown using a finger to make a circular rub-out in the drying paint.
To determine the influence of the additives on color compatibility and color strength, L*, a*, b*, c*, h* and ∆E* values were measured on the dried drawdowns using an X-Rite 939 Spectrodensitometer using a D65/10° illuminant/observer combination and an 8 mm measuring aperture. The tinted paint containing no post-added surfactant was used as the reference, and its color strength was set to 100%. Each drawdown was evaluated to determine: color strength after two and ten minutes of shaking, rub-out test after two and ten minutes of shaking, and ∆E between the drawdowns obtained after two and ten minutes of shaking. The ∆E* values less than 0.5 were considered to show no color difference; 0.5 < ∆E* < 1.0 were only slightly visible, while ∆E* values greater than 1.0 were deemed to be unacceptable.
Paint viscosities were measured using a Brookfield DV-I Prime viscometer, spindle 64 at 100 RPM. Sag resistance was determined using an Erichsen Sag Tester Model 419 (ASTM
D 4400 version).
Results
Initial screening studies were first conducted in order to determine whether any trends were immediately obvious as well as to get better ideas for future experimental direction. In these screenings, a broad number of surfactants and dispersants were post-added at 1.0 wt% to a base paint; once thoroughly mixed, the base paint was tinted using a designated dose of colorant. Because of the breadth of chemistries evaluated, top performers for a given base paint-colorant system could be identified; however, a systematic structure-property understanding was not possible.
For example, in a waterborne acrylic base paint that was tinted using 1.0 wt% of a pigment violet 23 colorant, it was observed that many surfactants of varying chemistries were able to slightly to significantly improve the color strength of the paint; however, only the alkoxylated acetylenic diol (TMDD-EOPO) showed a significant improvement (∆E < 0.5) in the rub-out test (Figure 2).
While there is a very slight improvement in both color strength and rub-out (∆E) when Benchmark C (a potassium alkyl phosphate ester) is added, the significant improvements observed when the TMDD-EOPO additive is used is noteworthy (Figure 3).
Because of the positive improvements in color acceptance that were seen for the TMDD-EOPO, additional screening tests were conducted to determine what effects type and degree of alkoxylation have on color strength and rub-out. To do this, a series of acetylenic diol ethoxylates (TMDD-4EO, TMDD-10EO and TMDD-23EO) were evaluated as 1.0 wt% post-additions to the same waterborne acrylic paint used in the previous study, and the color strength and rub-out data after tinting with 1.0 wt% pigment violet 23 colorant was compared to that obtained for the control (no additive) and TMDD-EOPO (Figure 4).
All the alkoxylated acetylenic diols significantly improve the color strength obtained on tinting the acrylic base paint with 1.0 wt% pigment violet 23 colorant. This is not surprising because it is well known that dynamic wetting agents can enable more efficient wetting of pigment particles in order to enable better dispersion of the pigment particles; as long as the polymeric dispersant present in the formulation can stabilize these smaller pigment particles, higher color strength results.6 As the degree of acetylenic diol ethoxylation decreases, the color strength increases. This correlates with previous observations that acetylenic diol ethoxylates with higher degrees of ethoxylation are less efficient at lowering dynamic surface tension than their low-mole ethoxylate analogues. The tendency of these highly branched wetting agents to resist being adsorbed on particle surfaces ensures that more of the molecules are available to provide this needed dynamic wetting benefit.8-10 Interestingly, within the series of three acetylenic diol ethoxylates, both color strength and rub-out (∆E) values improve to more significant extents as degree of ethoxylation decreases. That said, only TMDD-EOPO, which has a degree of ethoxylation in between that of TMDD-4EO and TMDD-10EO but also has been hydrophobically modified via propoxylation, is able to render this base paint robust enough to tinting with the PV23 colorant (i.e., rub-out ∆E < 0.5).
A similar study was conducted using the same waterborne acrylic base paint but tinting it with 1.0 wt% of a pigment red 112 colorant. The results are shown in Figure 5. Here, again, the simple presence of 1.0 wt% of the dynamic wetting agents is enough to provide a 15% to > 20% increase in color strength. However, the improvement in rub-out values varies with degree of alkoxylation. Interestingly, the additive with the highest level of ethoxylation (TMDD-23EO) provides only somewhat better colorant acceptance while lower levels of ethoxylation show even better colorant acceptance. Here there appears to be a “sweet spot,” with TMDD-10 (the ~ 10 mole ethoxylate) providing the optimal colorant acceptance.
In order to understand whether similar trends would translate to base paints and colorants of different chemistries, a waterborne styrene-acrylic base paint that was tinted using 1.0 wt% of a pigment blue 15:3 colorant was studied. Tinting this formulation with the PB15:3 colorant resulted in a slight rub-out difference (∆E ~ 0.6); however, the paint’s color strength could be improved significantly with the use of a wide variety of surfactants. That said, many of the additives tested, including the alkoxylated acetylenic diols, adversely impacted the colorant acceptance, resulting in ∆E values > 1 (Figure 6). In this system, a series of linear alcohol ethoxylates of varying hydrophile-lipophile balance (HLB) values was also studied.
While these alcohol ethoxylates represent a broad distribution of molecules exhibiting several different hydrophobic groups (i.e., of different carbon number) as well as varying degrees of ethoxylation, HLB values can be a useful way to express a surfactant’s overall physico-chemical characteristics. A surfactant with a higher HLB value is more hydrophilic in nature than those with lower HLB values.11
As shown in Figure 6, the base paint has good color acceptance already and only the highest HLB alcohol ethoxylate (AE HLB 18.2) can improve both the color strength and the rub-out (∆E) for this base paint-colorant system. This is likely due to the ability of AE HLB 18.2 to provide steric stabilization – acting as a co-dispersant – to improve the overall stabilization of the white pigments and fillers in the styrene-acrylate base paint. In such a formulation, the styrene-acrylic binder also functions as a polymeric dispersant for the particles, operating via an electrostatic stabilization mechanism.
Because the behavior of surfactants in base paints varies greatly depending on the chemical natures of the binder as well as the other components of the base paint, it was decided to conduct an evaluation of surfactants in a polyurethane/alkyd trim paint formulation in which most of the chemical composition could be understood. Therefore, the base paint formulation shown in Table 1 (Experimental) was prepared. In this formulation, the dry titanium dioxide was ground with a formulated dispersant that contains an anionic polymeric dispersant (providing electrostatic stabilization) as well as two nonionic surfactants (providing dynamic wetting and temporary steric stabilization). 1.0 wt% of each Color Acceptance Additive was post-added as in previous studies, and, finally, each base paint was tinted using 5.0 wt% carbon black colorant (PBk7). A comparison of color strength and rub-out (∆E) values is shown in Figure 7.
This base paint appears to be adequately stabilized already, but the addition of 1.0 wt% alcohol ethoxylate or alkoxylated acetylenic surfactant can still improve the color strength by 3-7% relative to the control (no additive). Interestingly, significant improvement in rub-out (∆E) values does not appear to strictly correlate with the HLB value for the alcohol ethoxylates. Moreover, many of these surfactants affect viscosity and sag resistance upon tinting, as can be seen in Figure 8. Therefore, an additive that improves color strength, rub-out and has less of an impact on viscosity and sag resistance would be the best option. In this regard, the Formulated Nonionic Surfactant and the AE HLB 18.2 appear to be the better choices amongst the surfactants studied here (Figure 9). Clearly the complex nature of coating formulations and the variety of interactions that are available to surface active molecules makes it rather difficult to develop a robust predictive model that can be used to identify the best additive for improving color acceptance without adversely affecting other properties.
Discussion
Color development in a tinted base paint is dependent upon the stability of the pigments and fillers dispersed in both the base paint and the colorants. When the colorants and paint are mixed, the additives present in each component can re-equilibrate between all pigments and fillers present in the formulation. Many base paints, such as the waterborne acrylic base paint studied here, use the minimum quantity of additives needed to stabilize the titanium dioxide and fillers in the base paint. Tinting these under-stabilized base paints often results in a re-distribution of additives – removing them from the colored pigments that come from the colorant in order to satisfy the additive-starved particles present in the base paint. Colorant destabilization and loss of color strength result (Figure 10).
The formulation of pigment dispersions used in both base paints and colorants can be enhanced by using the optimal surface active agents to wet, disperse and stabilize the solid particles in the medium as well as to provide improved performance of the paint during application. As can be seen from the composition of the dispersant used in the waterborne polyurethane/alkyd base paint in Table 1, most dispersions employ at least two, and often three or more, surface active components that are combined in order to achieve the desired properties. These surfactants can also help to improve the compatibility of the dispersion when it is letdown into another system. Similarly, they can help minimize shock and color acceptance issues when a colorant is added to a base paint during tinting. It is believed that these surfactants function by migrating to the unprotected pigment surfaces created when the dispersing additives that are originally protecting the pigment re-distribute on mixing. Thus, these surfactants can prevent the destabilization of the pigments and lead to excellent colorant acceptance (Figure 11).
Conclusions
The experiments conducted in this study have demonstrated that nonionic surfactants, like alkoxylated acetylenic diols, that lower dynamic surface tension are also often effective at increasing the color strength achievable on tinting a given base paint. Additionally, alcohol ethoxylates – particularly those with higher HLB values – are often able to improve color acceptance presumably because of their ability to provide steric stabilization to pigments and fillers. In this way, the high-mole alcohol ethoxylates can function as co-dispersants when used in combination with acrylic or styrene-acrylic binders that provide the needed anionic stabilization. All these additives can stabilize the pigments during surfactant/dispersant re-equilibration after mixing. However, some formulations see improvements in color acceptance when higher-mole alkoxylates are used while other paints require the more hydrophobic lower-mole alkoxylates to see such improvements. It is likely that the chemistries employed in both the base paint and the colorants play significant roles in determining what is needed to stabilize all particles in the combined formulation; therefore, predictability
remains elusive.
References
1. S. Heutz, M.N.S. Kunnen and B.A. Wilbanks, Customized Tinting System Design: Advanced Tinting Systems – An Integrated Concept, Proceedings of the 39th Annual Waterborne High-Solids, and Powder Coating Symposium, 2012, DEStech Publications, Inc., Lancaster, PA, 617-627.
2. M. Kunnan and S. Heutz, Integrated Tinting Systems, Paint and Coatings Industry, 2011 (http://www.pcimag.com/articles/93432-integrated-tinting-systems).
3. Fluid Management Inc., Wheeling, IL, USA (http://www.fluidman.com).
4. COROB S.p.A., San Felice Sul Panaro MO, Italy (http://www.corob.com).
5. C. Louis, Improving Colourant Compatibility and Colour Development in Architectural Coatings, Pitture e Vernici - European Coatings – Formulation, 2016, 5, 22-28.
6. K.M. Peck, Formulating Optimized Aqueous Dispersions Using Surface Active Additive Triangulation, Journal of Coatings Technology, 2014, 50-57.
7. K.M. Peck, The Role of Surfactants in Aqueous Pigment Dispersion, Paint and Coatings Industry, 2016, 62-66.
8. J. Schwartz, The Importance of Low Dynamic Surface Tension in Waterborne Coatings, Journal of Coatings Technology, 1992, 64, 65-74.
9. S.W. Musselman and S. Chander, Adsorption of Acetylenic Diol-Based Nonionic Surfactants on Lampblack and Phthalocyanine Blue Pigment, Journal of Colloid and Interface Science, 2002, 256, 91-99.
10. J.A. Marsella, C.J. Mammarella and I.K. Meier, Interactions of surfactants with a derivatized low molecular weight styrene maleic anhydride co-polymer – differences between acetylenic diol-based wetting agents and other ethoxylates, Colloids and Surfaces A: Physicochem. Eng. Aspects, 2004, 234, 63-69.
11. W.C. Griffin, Classification of Surface-Active Agents by “HLB”, Journal of the Society of Cosmetic Chemists, 1949, 5, 311-326.