With recent EPA and ECHA interest in PFAS compoundsi and impending new restrictions, many end users are currently searching for alternatives to fluoroalkyl based products. This is not trivial as PFAS offer unique properties and performances. Beyond their exceptional properties, their much higher costs make it probable that these products were adopted in the applications for strong reasons and are not going to be easily replaced.
Over the many years that these have been available, the use of Perfluoro Alkyl Substance (PFAS) has found utility in a plethora of markets and applicationsii. These can be logically divided into physical performance categories such as surface tension/energy; COF, hydro and oleophobicity, stain and chemical resistance.
We at Siltech believe that PFAS are an extremely unique and proven useful family of polymers. We are not taking a side in this issue, but rather offering our specialty silicone expertise for those who choose to reformulate. What follows are some comparisons of how organosilicones equate to PFAS compounds in the critical performance criteria above.
One word of caution, aqueous use levels tend to be an order of magnitude higher with organosilicones relative to PFAS. The use levels are still very low compared to organic alternatives. The lower costs of silicone products offset the economics, but using more can make undesirable side effects more likely.
In the very important Aqueous Fire Fighting Foams (AFFF) we have shown improved coefficient of spreading in a customer supplied AFFF formulation relative to heptanesiii. Most of the silicone surfactants retained the foam, clarity and aesthetics of the PFAS control while dramatically improving the spreading coefficient. The data shows that the silicone surfactants are worthy of further consideration in this critical surface forces driven application currently dominated by PFAS surfactants.
In a classic approach, we achieve this level of water repellency with silicone quaternary ammonium compounds. These materials can also offer quick exhaustion onto glass, textiles or other surfaces from solution. Some of these dialkyl quaternary compounds have an added feature as they can be effective as antiseptics; although significant regulatory hoops need to be navigated to claim this usage in a product.
In what is likely the simplest approach, basic film forming silicone emulsions give an effective, if not exciting, 80° contact angle on glass. This number is artificially low due to the emulsifiers and improves as the emulsifiers are rinsed out of the network. Delivery of very similar materials from solvent yields 115° contact angle on glass. Also, these materials are extensively used, especially in roof and concrete coatings and leather treatments, so in the real world they are giving effective repellency. The high cost-effectiveness and simplicity of these materials makes them excellent choices for water repellency.
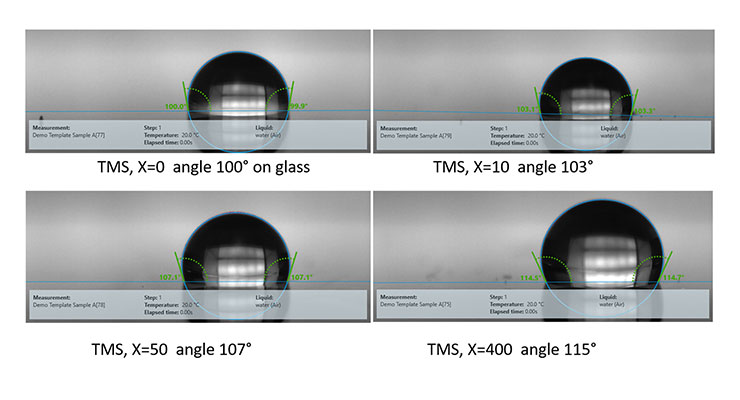
Another approach uses Silmer® TMS materials which have trimethoxy silane (TMS) functionality on the silicone polymer. These orient especially well to reactive surfaces such as glass, textiles and metals providing up to 115° contact angles. There is no alkyl chain in this case, but the chain length of the silicone increases hydrophobicity and provides equivalent performance to the silicone quat approach above. This approach works quite well alone but is also shown to improve the performance of the simple film forming emulsions.
The most recent approach to water repellency is offered by Q resin containing silicon species. The highly cross linked Silmer Q resins can be delivered from solvent, in a sol-gel formulation or from an emulsion. These approaches also give 115° contact angles on glass.
Beyond contact angles, we utilized AATCC 193 to evaluate water repellency of treated cotton fabric swatches. This protocol uses blends of water/IPA with increasing ratios of IPA - and therefore lower ST - to wet out the swatches. Each solution is dyed for simplicity. The cotton swatches are treated with the test solutions and heated briefly to dry them. They are then evaluated for beading of the standard aqueous/IPA solutions.
The test solutions are 100% RO water (colorless, 72 mN/m); 2% IPA (blue, 59 mN/m); 5% IPA (Pink, 50 mN/m); 10% IPA (Orange, 42 mN/m); 20% IPA (Yellow, 33 mN/m); and 30% IPA (Dark Blue, 18 mN/m). Grades are assigned of the lowest ST solution to still bead on the fabric according to 0 (colorless) - 5 (Dk. Bl.).
Our best formulation, containing Q resins beat an over-the-counter product which contains a PFAS. The PFAS control rated a 3 with clear beading in the orange (42 mN/m) line of drops but wet through of the 33 mN/m yellow column. Our QT Silicon Resin with an aminosilicone delivered as a sol-gel exceeds this, rating an easy 4. In that sample even the most stringent dark blue 28 mN/m solution is only partially wetting the treated fabric.
We have found and published previously that linear organosilicone materials with chemical anchors on the organic appendages offer some oleophobicityv,vi, as measured by stain resistance. While the results are not at the level of PFAS compounds there is significant performance improvement, often comparable to our perfluorobutyl (~C4F9) based fluoroalkyl silicone products.
Expanding on that work, we treated plugs made from a customer’s proprietary grout with 5% of various organosilicones using a PFAS treated control. These were evaluated with water and sunflower oil and then for stain repellency. While many showed hydrophobicity, an amino-functional film forming emulsion; a silicone film forming emulsion; and a Silmer TMS emulsion blended with the amino-functional emulsion showed Sunflower oil contact angles comparable to the PFAS treated tiles. This oleophobicity is not generally seen, but demonstrates in some applications the orientation of the silicone at the surface can provide oleophobicity.
In order to evaluate the stain repellency, the grout tiles were treated with ketchup, mustard, red wine vinegar, balsamic vinegar, instant coffee and soya sauce. Stain resistance before and after rinse off was measured using Nix Pro 2 Color Sensor in terms of CIELAB data: L*, a* and b*. L*, a* and b* are the coordinates in Lab colour space. Measuring the L*, a* and b* data before and after staining, one can calculate Delta E, which is the distance between two colours designated as two points in the Lab colour space. A smaller value for Delta E tells that the color change before and after staining is small. A sample with a smaller Delta E value has better stain resistance compared to a sample with a bigger Delta E valuevii.
Three samples show results quite close to the PFAS fortified plugs. The three are film forming emulsions – one combined with a Silmer TMS products. Averaged across all of the silicone samples the soya sauce, vinegar and ketchup were well controlled while the coffee and mustard were more difficult but still close to the PFAS in the best samples.
In a separate technical approach to evaluate oleophobicity, we have found that some high Tg modifications, either hydrocarbon wax or EO chains, provide oil protection at room temperature. In these experiments, we treated sample cardboard sections with a series of Silsurf® Silicone Polyether derivatives or Silwax® hydrocarbon modified silicones.
A few drops of vegetable oil were placed on the treated cardboard samples and evaluated over an hour at ambient conditions. The control and many of the treated cardboard sections absorbed the vegetable oil over the hour.
However, the sample which has a waxy hydrocarbon chain and is a high melting wax itself gave very good beading of the vegetable oil after one hour. Similarly, the sample which has a waxy EO chain and is a low melting wax (~40°C) also shows strong beading with no absorption after the hour. One current solution for food packaging is high MW waxes which are believed to physically block absorption, that is a possible explanation for these results.
The cookware industry has replaced Teflon® coated cookware with ceramic coated pots and pans for effective oleophobicity, heat resistance and release. Ceramics are silicon-based matrices highly cross-linked at very high temperatures. Following this idea, we wondered how our Q resin materials would fare. These also react in all four directions offering cross-link densities in the direction of those found in high heat cured ceramics. While this is a nascent area of research, we are intrigued by the early results.
Silmer DTQ-75 resin in the same cardboard treatment protocol used above gave strong beading after an hour, with no visible absorption. This is not a waxy product and so must be due to the high cross-link density of this material.
As a final example, a brand-new and proprietary composition developed in our labs has also shown oleophobic efficacy. This UV reactive substance is combined with an acrylate functional UV resin and cured onto an Aluminum panel.
Droplets of white mineral oil applied to the surfaces of the UV cured control (no additive) and this product show a distinct difference in beading.
While silicone itself is pretty good at heat resistance, the Si-O-Si bonds are very labile to chemical degradation in an acid/base sense. One could look at it that the strong C-F stability is why PFAS tend to build up in the environment and the hydrolytic lability of PDMS is its main mechanism of degradation in the environmentviii.
The PDMS base polymer is thermally stable to about 150°C. However, adding organic groups to the silicone to gain solubility, reactivity, and other properties reduces the thermal stability to that of the organic moiety. In other words, the PDMS does nothing to stabilize the heterolysis of C-H bonds in the organic appendages.
Regarding chemical resistance, we are again looking to Q resins to improve chemical stability. These Q resins have less organic functionality and are highly hindered to SN2 acid/base chemistry. This hindrance to nucleophilic attack results in alkaline stability at least.
Leatherman et.al.ix published carbosilane analogues to PDMS surfactants. This is elegant work which I will simplistically describe as replacing the O of PDMS with CH2 groups. Because the hydrolytic lability of siloxane polymers is due to breaking the Si-O-Si bonds, this invention creates an acid/base stable polymer with very similar surface energy properties to PDMS derivatives.
These two methods are the only general approaches we envision to address the chemical resistance of PFAS with silicon-based materials. This may be a situation where organic surfactants, if their higher surface energies can be used, are the better option.
We have multiple strong solutions to provide hydrophobicity, wetting, aqueous stain repellency and stabilization properties from multiple silicon-based approaches. For oleophobicity and lipid stain resistance we have demonstrated early results with three approaches and are anxious to work on this further.
Regarding chemical stability, especially in pH extreme aqueous milieu this is one area where silicone solutions are not likely to be part of the solution. The easy hydrolysis of silicone backbones, which allows them to chemically degrade in the environment, is in stark contrast to the C-F bond structures of PFAS that are inert to these environments.
Siltech is anxious to work directly with anyone who would like to evaluate our technologies in these or any problems. Your Technology - Our Chemistry is more than a motto; it is an invitation.
ii Glüge, et.al. Environ. Sci.: Processes Impacts, 2020, 22, 2345-2373
iii Ruckle, et.al. Proceedings of the 51st Waterborne Symposium, 2024, University of Southern Mississippi.
iv Ruckle, et.al. Proceedings of the 50th Waterborne Symposium, 2023, University of Southern Mississippi.
v Ruckle, et.al. “Novel Organosilicone Fluoro-Free Anti-Graffiti Agents”, Proceedings of the Waterborne Symposium, 2016, University of Southern Mississippi.
vi Ruckle et.al. “Fluoro-Free Anti-Graffiti Properties from A Novel OrganoSilicone”, European Coatings Show (2017)
vii ∆E=√(∆L^*)2 + (∆a*)2 + (∆b*)2; ∆L* = ∆L* (after stain)
– ∆L* (before stain)
viii Graiver, D., “Farminer, K.W. & Narayan, R. A Review of the Fate and Effects of Silicones in the Environment”. Journal of Polymers and the Environment 11, 129–136 (2003).
ix Leatherman, et.al. US 7,700,797, (2010)
Over the many years that these have been available, the use of Perfluoro Alkyl Substance (PFAS) has found utility in a plethora of markets and applicationsii. These can be logically divided into physical performance categories such as surface tension/energy; COF, hydro and oleophobicity, stain and chemical resistance.
We at Siltech believe that PFAS are an extremely unique and proven useful family of polymers. We are not taking a side in this issue, but rather offering our specialty silicone expertise for those who choose to reformulate. What follows are some comparisons of how organosilicones equate to PFAS compounds in the critical performance criteria above.
Surface Forces
In many of the surface applications, organofunctional silicones are an excellent option for replacing a PFAS. The silicone polymer used as a backbone in the chemical hybrids of silicone and organic moieties has a surface energy of 20 mN/m. This is only “beaten” by PFAS compounds at 13-20 mN/m, in other words there are no common surfaces that need less than 20 mN/m to be wetted. Also, we chemically append hydrophiles to the silicone polymer giving surfactants which reduce the surface tension of aqueous systems as low as 20.5 mN/m at 0.1%. These can be designed to control or stabilize foam as well.One word of caution, aqueous use levels tend to be an order of magnitude higher with organosilicones relative to PFAS. The use levels are still very low compared to organic alternatives. The lower costs of silicone products offset the economics, but using more can make undesirable side effects more likely.
In the very important Aqueous Fire Fighting Foams (AFFF) we have shown improved coefficient of spreading in a customer supplied AFFF formulation relative to heptanesiii. Most of the silicone surfactants retained the foam, clarity and aesthetics of the PFAS control while dramatically improving the spreading coefficient. The data shows that the silicone surfactants are worthy of further consideration in this critical surface forces driven application currently dominated by PFAS surfactants.
Coefficient of Friction (COF)
COF likely results directly from surface energy, but it is important enough to briefly discuss. We have seen silicone materials lower COF to 0.150 which is likely a minimum. With organosilicone structures which have been designed to minimize COF, one typically obtains COF values at or below 0.200.Water Repellency
We have recently published a review of different methods for generating water repellencyiv. While these approaches all achieve about 115° contact angle on glass, each has other strengths and weaknesses.In a classic approach, we achieve this level of water repellency with silicone quaternary ammonium compounds. These materials can also offer quick exhaustion onto glass, textiles or other surfaces from solution. Some of these dialkyl quaternary compounds have an added feature as they can be effective as antiseptics; although significant regulatory hoops need to be navigated to claim this usage in a product.
In what is likely the simplest approach, basic film forming silicone emulsions give an effective, if not exciting, 80° contact angle on glass. This number is artificially low due to the emulsifiers and improves as the emulsifiers are rinsed out of the network. Delivery of very similar materials from solvent yields 115° contact angle on glass. Also, these materials are extensively used, especially in roof and concrete coatings and leather treatments, so in the real world they are giving effective repellency. The high cost-effectiveness and simplicity of these materials makes them excellent choices for water repellency.
Another approach uses Silmer® TMS materials which have trimethoxy silane (TMS) functionality on the silicone polymer. These orient especially well to reactive surfaces such as glass, textiles and metals providing up to 115° contact angles. There is no alkyl chain in this case, but the chain length of the silicone increases hydrophobicity and provides equivalent performance to the silicone quat approach above. This approach works quite well alone but is also shown to improve the performance of the simple film forming emulsions.
The most recent approach to water repellency is offered by Q resin containing silicon species. The highly cross linked Silmer Q resins can be delivered from solvent, in a sol-gel formulation or from an emulsion. These approaches also give 115° contact angles on glass.
Beyond contact angles, we utilized AATCC 193 to evaluate water repellency of treated cotton fabric swatches. This protocol uses blends of water/IPA with increasing ratios of IPA - and therefore lower ST - to wet out the swatches. Each solution is dyed for simplicity. The cotton swatches are treated with the test solutions and heated briefly to dry them. They are then evaluated for beading of the standard aqueous/IPA solutions.
The test solutions are 100% RO water (colorless, 72 mN/m); 2% IPA (blue, 59 mN/m); 5% IPA (Pink, 50 mN/m); 10% IPA (Orange, 42 mN/m); 20% IPA (Yellow, 33 mN/m); and 30% IPA (Dark Blue, 18 mN/m). Grades are assigned of the lowest ST solution to still bead on the fabric according to 0 (colorless) - 5 (Dk. Bl.).
Our best formulation, containing Q resins beat an over-the-counter product which contains a PFAS. The PFAS control rated a 3 with clear beading in the orange (42 mN/m) line of drops but wet through of the 33 mN/m yellow column. Our QT Silicon Resin with an aminosilicone delivered as a sol-gel exceeds this, rating an easy 4. In that sample even the most stringent dark blue 28 mN/m solution is only partially wetting the treated fabric.
Oleophobicity
Perhaps the most unique thing that PFAS do is repel oils. Oil-based stain resistance is important in construction membranes, solar cells, petroleum transport and storage, chemical and plastics processing, textiles and PPE, leather finishes, paper manufacture, sealants, sporting goods, and other areas. An especially big need for this property is lipstick, motor oil, and grease resistance of textiles and especially for food wrappers, carpet and textile treatments and PPE. There are a lot of things that can repel water effectively, but repelling oils is difficult. Simple organosilicone treatments are usually relatively ineffective at providing this oleophobicity.We have found and published previously that linear organosilicone materials with chemical anchors on the organic appendages offer some oleophobicityv,vi, as measured by stain resistance. While the results are not at the level of PFAS compounds there is significant performance improvement, often comparable to our perfluorobutyl (~C4F9) based fluoroalkyl silicone products.
Expanding on that work, we treated plugs made from a customer’s proprietary grout with 5% of various organosilicones using a PFAS treated control. These were evaluated with water and sunflower oil and then for stain repellency. While many showed hydrophobicity, an amino-functional film forming emulsion; a silicone film forming emulsion; and a Silmer TMS emulsion blended with the amino-functional emulsion showed Sunflower oil contact angles comparable to the PFAS treated tiles. This oleophobicity is not generally seen, but demonstrates in some applications the orientation of the silicone at the surface can provide oleophobicity.
In order to evaluate the stain repellency, the grout tiles were treated with ketchup, mustard, red wine vinegar, balsamic vinegar, instant coffee and soya sauce. Stain resistance before and after rinse off was measured using Nix Pro 2 Color Sensor in terms of CIELAB data: L*, a* and b*. L*, a* and b* are the coordinates in Lab colour space. Measuring the L*, a* and b* data before and after staining, one can calculate Delta E, which is the distance between two colours designated as two points in the Lab colour space. A smaller value for Delta E tells that the color change before and after staining is small. A sample with a smaller Delta E value has better stain resistance compared to a sample with a bigger Delta E valuevii.
Three samples show results quite close to the PFAS fortified plugs. The three are film forming emulsions – one combined with a Silmer TMS products. Averaged across all of the silicone samples the soya sauce, vinegar and ketchup were well controlled while the coffee and mustard were more difficult but still close to the PFAS in the best samples.
In a separate technical approach to evaluate oleophobicity, we have found that some high Tg modifications, either hydrocarbon wax or EO chains, provide oil protection at room temperature. In these experiments, we treated sample cardboard sections with a series of Silsurf® Silicone Polyether derivatives or Silwax® hydrocarbon modified silicones.
A few drops of vegetable oil were placed on the treated cardboard samples and evaluated over an hour at ambient conditions. The control and many of the treated cardboard sections absorbed the vegetable oil over the hour.
However, the sample which has a waxy hydrocarbon chain and is a high melting wax itself gave very good beading of the vegetable oil after one hour. Similarly, the sample which has a waxy EO chain and is a low melting wax (~40°C) also shows strong beading with no absorption after the hour. One current solution for food packaging is high MW waxes which are believed to physically block absorption, that is a possible explanation for these results.
The cookware industry has replaced Teflon® coated cookware with ceramic coated pots and pans for effective oleophobicity, heat resistance and release. Ceramics are silicon-based matrices highly cross-linked at very high temperatures. Following this idea, we wondered how our Q resin materials would fare. These also react in all four directions offering cross-link densities in the direction of those found in high heat cured ceramics. While this is a nascent area of research, we are intrigued by the early results.
Silmer DTQ-75 resin in the same cardboard treatment protocol used above gave strong beading after an hour, with no visible absorption. This is not a waxy product and so must be due to the high cross-link density of this material.
As a final example, a brand-new and proprietary composition developed in our labs has also shown oleophobic efficacy. This UV reactive substance is combined with an acrylate functional UV resin and cured onto an Aluminum panel.
Droplets of white mineral oil applied to the surfaces of the UV cured control (no additive) and this product show a distinct difference in beading.
Chemical Stability
Being in the upper-right corner of the periodic table, just left of the inert gas column, no element wants an electron more than the fluorine atom! This provides extremely strong bond strength to the C-F linkage which make PFAS very stable to chemical and thermal degradation. Typical applications that depend on this property are electronics, electroplating, chemical processing, aerospace, batteries, lubricants, paper bleaching, petroleum processing and transport.While silicone itself is pretty good at heat resistance, the Si-O-Si bonds are very labile to chemical degradation in an acid/base sense. One could look at it that the strong C-F stability is why PFAS tend to build up in the environment and the hydrolytic lability of PDMS is its main mechanism of degradation in the environmentviii.
The PDMS base polymer is thermally stable to about 150°C. However, adding organic groups to the silicone to gain solubility, reactivity, and other properties reduces the thermal stability to that of the organic moiety. In other words, the PDMS does nothing to stabilize the heterolysis of C-H bonds in the organic appendages.
Regarding chemical resistance, we are again looking to Q resins to improve chemical stability. These Q resins have less organic functionality and are highly hindered to SN2 acid/base chemistry. This hindrance to nucleophilic attack results in alkaline stability at least.
Leatherman et.al.ix published carbosilane analogues to PDMS surfactants. This is elegant work which I will simplistically describe as replacing the O of PDMS with CH2 groups. Because the hydrolytic lability of siloxane polymers is due to breaking the Si-O-Si bonds, this invention creates an acid/base stable polymer with very similar surface energy properties to PDMS derivatives.
These two methods are the only general approaches we envision to address the chemical resistance of PFAS with silicon-based materials. This may be a situation where organic surfactants, if their higher surface energies can be used, are the better option.
Summary
We think that the parallels in surfacy energy, incompatibility, and interfacial tension between PFAS and PDMS based products often make them the “next best thing” if one is feeling the regulatory squeeze on PFAS ingredients. This includes the critical AFFF formulations, where it is also likely that low surface tension Silsurf® surfactant could be part of future, non-PFAS solutions.We have multiple strong solutions to provide hydrophobicity, wetting, aqueous stain repellency and stabilization properties from multiple silicon-based approaches. For oleophobicity and lipid stain resistance we have demonstrated early results with three approaches and are anxious to work on this further.
Regarding chemical stability, especially in pH extreme aqueous milieu this is one area where silicone solutions are not likely to be part of the solution. The easy hydrolysis of silicone backbones, which allows them to chemically degrade in the environment, is in stark contrast to the C-F bond structures of PFAS that are inert to these environments.
Siltech is anxious to work directly with anyone who would like to evaluate our technologies in these or any problems. Your Technology - Our Chemistry is more than a motto; it is an invitation.
References
i https://www.epa.gov/newsreleases/epa-announces-new-drinking-water-health-advisories-pfas-chemicals-1-billion-bipartisanii Glüge, et.al. Environ. Sci.: Processes Impacts, 2020, 22, 2345-2373
iii Ruckle, et.al. Proceedings of the 51st Waterborne Symposium, 2024, University of Southern Mississippi.
iv Ruckle, et.al. Proceedings of the 50th Waterborne Symposium, 2023, University of Southern Mississippi.
v Ruckle, et.al. “Novel Organosilicone Fluoro-Free Anti-Graffiti Agents”, Proceedings of the Waterborne Symposium, 2016, University of Southern Mississippi.
vi Ruckle et.al. “Fluoro-Free Anti-Graffiti Properties from A Novel OrganoSilicone”, European Coatings Show (2017)
vii ∆E=√(∆L^*)2 + (∆a*)2 + (∆b*)2; ∆L* = ∆L* (after stain)
– ∆L* (before stain)
viii Graiver, D., “Farminer, K.W. & Narayan, R. A Review of the Fate and Effects of Silicones in the Environment”. Journal of Polymers and the Environment 11, 129–136 (2003).
ix Leatherman, et.al. US 7,700,797, (2010)